Cathepsin G Antibodies
Your search for reliable Cathepsin G antibodies ends here. Cathepsin G, known by aliases such as CTSG, Ctsg, LOC505658, LOC509956, ctsg, LOC100053921, LOC102174648, LOC100733619, LOC100154047, is an integral part of our antibody range. Whether you're working with Human, Rat, Mouse, Guinea Pig, or other species, our range of Cathepsin G antibodies offer precise detection across diverse samples. These specialized antibodies are tailored for various scientific applications like WB, ELISA, IHC, IF (cc), IF (p), providing you with options like polyclonal, recombinant, and monoclonal antibodies, sourced from different host species such as Rabbit, Mouse. The efficacy of our antibodies is well-established, demonstrated through multiple methods.
Detailed information, including references, images, and validations by other customers, can be found on each product page. Should you require assistance in finding a specific product, our customer service team is ready to assist. Utilize our Cathepsin G antibodies in your research endeavors for dependable Cathepsin G detection.
CTSG Reactivity: Human WB, IHC Host: Rabbit Polyclonal unconjugated
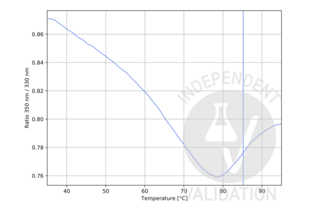
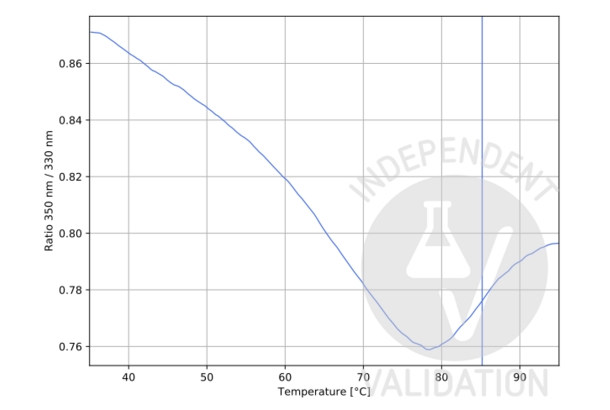 Cathepsin G antibody (Internal Region) (ABIN3183694)
Cathepsin G antibody (Internal Region) (ABIN3183694)
CTSG Reactivity: Human WB, ELISA Host: Rabbit Polyclonal unconjugated
CTSG Reactivity: Human WB, ELISA, ICC, IHC (p), IHC (fro) Host: Rabbit Polyclonal unconjugated
Cathepsin G Antibodies by Reactivity
Find Cathepsin G Antibodies for a variety of species such as anti-Human Cathepsin G, anti-Rat Cathepsin G, anti-Mouse Cathepsin G. The species listed below are among those available. Click on a link to go to the corresponding products.
Cathepsin G Antibodies by Host
Find Cathepsin G Antibodies with a specific Host. The Host listed below are among those available. Click on a link to go to the corresponding products.
Cathepsin G Antibodies by Clonality
Find available monoclonal or polyclonal Cathepsin G Antibodies. Click on a link to go to the corresponding products.
Popular Cathepsin G Antibodies
- (7)
- (1)
- (1)
- (5)
- (5)
- (4)
- (3)
- (3)
- (2)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
Latest Publications for our Cathepsin G Antibodies
: "IL-1 and IL-36 are dominant cytokines in generalized pustular psoriasis." in: The Journal of allergy and clinical immunology, Vol. 140, Issue 1, pp. 109-120, (2017) (PubMed).Aliases for Cathepsin G Antibodies
cathepsin G (CTSG) Antibodiescathepsin G (Ctsg) Antibodies
cathepsin G (LOC505658) Antibodies
cathepsin G (LOC509956) Antibodies
cathepsin G (ctsg) Antibodies
cathepsin G (LOC100053921) Antibodies
cathepsin G (LOC102174648) Antibodies
cathepsin G (LOC100733619) Antibodies
cathepsin G (LOC100154047) Antibodies
CATG Antibodies
CG Antibodies
CTSG Antibodies
Ctsg Antibodies
Did you look for something else?
- Cathepsin F Antibodies
- Cathepsin E Antibodies
- Cathepsin D Antibodies
- Cathepsin B Antibodies
- Cathepsin 7 Antibodies
- Cathelicidin Antibodies
- Catechol-O-Methyltransferase Antibodies
- Catalase Antibodies
- CASZ1 Antibodies
- CASS4 Antibodies
- CASR Antibodies
- CASQ2 Antibodies
- Caspase Activity and Apoptosis Inhibitor 1 Antibodies
- Caspase 9 Antibodies
- Caspase 8 Antibodies
- Caspase 7 Antibodies
- Caspase 6 Antibodies
- Caspase 4 Antibodies
- Caspase 3 Antibodies
- Caspase 2 Antibodies
- Cathepsin H Antibodies
- Cathepsin K Antibodies
- Cathepsin L1 Antibodies
- Cathepsin L2 Antibodies
- Cathepsin S Antibodies
- Cathepsin W Antibodies
- Cathepsin Z Antibodies
- CATSPER1 Antibodies
- CATSPER2 Antibodies
- CATSPER3 Antibodies
- CATSPER4 Antibodies
- CATSPERB Antibodies
- CATSPERG Antibodies
- Caudal Type Homeobox 1 Antibodies
- Caveolin 2 Antibodies
- Caveolin 3 Antibodies
- Caveolin-1 Antibodies
- CAZIP Antibodies
- CBFA2T2 Antibodies
- CBFA2T3 Antibodies




