Fab Fragment Antibodies
Categories
Fab fragment antibodies (Fab = fragment antigen-binding region) are a valuable tool in molecular biology research and diagnostics. These antibodies are generated through the process of papain digestion, which cleaves the antibody molecule and produces Fab fragments. The Fab fragments, with a molecular weight of approximately 50 kDa, retain the antigen-binding regions of the original antibody while lacking the Fc (Fc = fragment crystallizable region) portion.
Discover our range of Fab Antibodies
By removing the Fc portion, Fab fragment antibodies eliminate the potential for interactions with Fc receptors found on cells or in vitro Fc-binding mechanisms. This feature makes Fab fragments particularly suitable for studies that require precise targeting of specific antigens without interference from Fc-mediated processes.
One of the primary applications of Fab fragment antibodies is in experiments where two primary antibodies from the same host species are used, such as two mouse monoclonal antibodies of the IgG isotype. In such cases, a conjugated Fab fragment anti-mouse IgG is applied first. This Fab fragment antibody binds specifically to the surface of the mouse IgG antibody, effectively blocking all available binding sites for anti-mouse IgG antibodies.
The monovalent nature of Fab fragment antibodies prevents them from binding to additional mouse IgG antibodies even if reintroduced into the system. This property is crucial in subsequent steps of the experiment. Following the application of the conjugated Fab fragment antibody, a second primary antibody (also a mouse IgG) is applied, targeting a different antigen of interest. To detect the presence of the second primary antibody, a second conjugated anti-mouse IgG is used.
Fab fragment antibodies offer numerous advantages in experimental settings where multiple primary antibodies from the same host species are used. Their monovalent nature, combined with the ability to block binding sites, provides a means to overcome challenges in detecting and differentiating target antigens. With their optimized permeability, Fab fragment antibodies have become indispensable tools in various research applications, including immunohistochemistry, immunofluorescence, and flow cytometry.
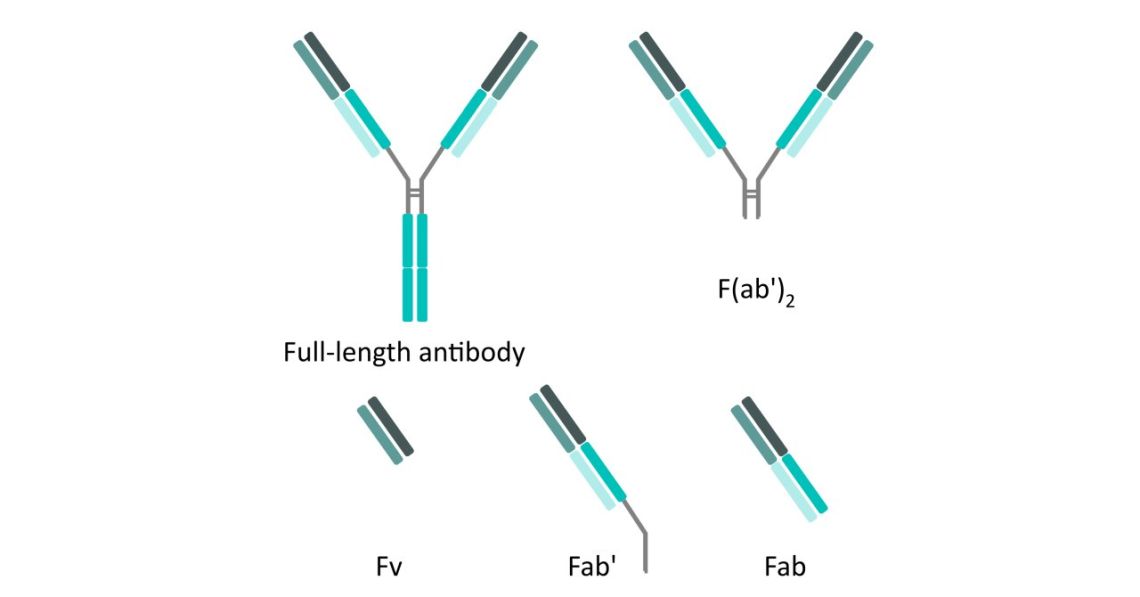
Figure 1. Fab fragment antibodies: Structure of full-length antibody, F(ab')2, Fv, Fab' and Fab fragments
Featured Fab Antibodies
F(ab')2 Fragment Secondary Antibodies
F(ab')2 fragment secondary antibodies provide an effective solution for minimizing non-specific binding interactions between the Fc portions of antibodies and Fc receptors on cells. These antibodies are generated through the process of pepsin digestion, which selectively removes most of the Fc region while preserving the hinge region. As a result, F(ab')2 fragments retain two antigen-binding F(ab) portions linked together by disulfide bonds, giving them a molecular weight of approximately 110 kDa.
F(ab')2 Fragment Secondary Antibodies
The main advantage of using F(ab')2 fragment antibodies is their ability to eliminate unwanted binding to Fc receptors. This is particularly important when working with tissues or cells that express Fc receptors, such as spleen, blood cells, hematopoietic cells, and leukocytes. By using F(ab')2 fragments, non-specific interactions between the Fc region of the antibody and Fc receptors on these cells are significantly reduced or completely eliminated.
F(ab')2 fragment antibodies are commonly employed in various experimental techniques, including flow cytometry, immunohistochemistry, and immunofluorescence. Their application as conjugated secondary antibodies allows for specific and reliable detection of target antigens in these assays. The F(ab')2 fragment conjugated secondary antibodies can be conveniently paired with primary antibodies raised in different species, facilitating the detection and visualization of specific antigens in complex biological samples.
Flow cytometry, a powerful technique for analyzing and sorting cells based on their surface markers, benefits from the use of F(ab')2 fragment conjugated secondary antibodies. By choosing an F(ab')2 fragment, researchers can minimize non-specific binding to Fc receptors, leading to improved specificity and accuracy in identifying and characterizing cell populations.
In immunohistochemistry, which involves the localization and visualization of antigens within tissue sections, F(ab')2 fragment conjugated secondary antibodies play a crucial role. Their reduced interaction with Fc receptors minimizes background staining and enhances the signal-to-noise ratio, resulting in clearer and more reliable staining patterns.
Similarly, in immunofluorescence, where fluorescent labels are used to visualize the distribution and co-localization of proteins in cells or tissues, F(ab')2 fragment conjugated secondary antibodies provide enhanced specificity and sensitivity. By using F(ab')2 fragments, researchers can avoid unwanted binding to Fc receptors, leading to improved accuracy in detecting the target antigens and reducing background fluorescence.
F(ab')2 fragment secondary antibodies offer a valuable tool for researchers aiming to minimize non-specific binding interactions and improve the specificity of their experiments. With their intact antigen-binding regions and reduced Fc-mediated effects, F(ab')2 fragment antibodies are particularly suitable for applications involving tissues or cells with Fc receptors. Their application in flow cytometry, immunohistochemistry, and immunofluorescence assays enables precise and reliable detection of target antigens, enhancing the quality and accuracy of experimental results.




