The magnetic beads technology has a lot of application fields. It can be utilized for the manipulation of biological material, including cells, nucleic acids, proteins microorganisms. Magnetic Cell Separation, also known as Magnetic Activated Cell Sorting is accomplished on the basis of certain surface structures of the cells, in contrast to common separation techniques like like filtration or centrifugation. The beads can be further used in following applications1:
- As carriers of antigens, antibodies, catalyzers, proteins and nucleic acids for tissue specific targeting
- Magnetic Resonance Imaging and Magnetic Particle Imaging
- Nanomaterial-based catalysis
- CUT&RUN and CUT&Tag
Workflow
The easy and efficient collection of beads in magnetic fields allows for easy rinsing and removal of excess reagents and ligand after coupling the ligand molecule, as well as usage in downstream applications.This method does not require columns or centrifugation steps, and are therefore ideal in high-throughput and automated applications. The general workflow can be summarized in three general steps (fig.1):
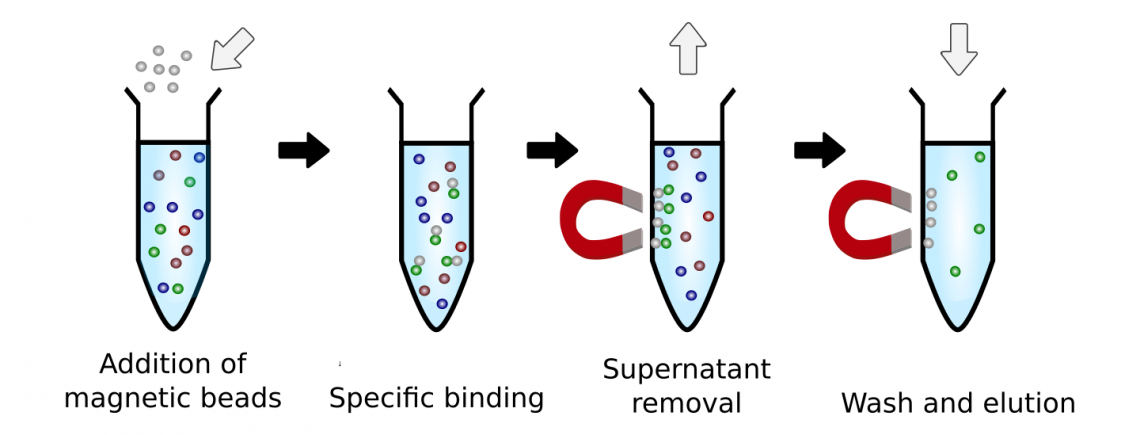
Bind
Add the binding buffer of choice to the sample and mix well to get a homogeneous suspension. Add beads. Mix beads by vortexing before adding them to the sample. Mix sample and incubate to enable the binding of bead and molecule of interest.
Wash
The beads respond to a field in a magnetic separator allowing bound material to be rapidly and efficiently separated from the rest of the sample. Unbound material is simply removed by aspiration, and the bead-bound target washed by the use of the magnet. At least two wash steps are recommended.
Elute
The bead-bound target is released in a suitable volume for use in downstream applications. Alternatively, the bead-bound target can be used directly while still attached to the beads.
Discover our Beads & Resin
- Compare and order our bead-based products!
Bead Design
Different surface modifications and bead sizes allow for choosing the optimal product for the right molecule to be coupled.
Magnetic Core
Magnetic beads used in purification contain a magnetic core which is covered in different materials. They are typically either Ferrimagnetic, or superparamagnetic3(fig.2).
Ferrimagnetic magnetic cores are with over 30 nm relatively large with a strong magnetic moment. They retain this magnetic moment even after removal of the magnetic field. The strong magnetic field leads to a fast separation of the beads in the magnetic field. Possible downfalls are self-magnetism and unwanted attachment to metal surfaces.
Superparamagnetic cores appear, depending on materials, in particles below 50 nm with a single magnetic domain. These single-domain magnets align all their magnetic moments in the same direction, so that when an external magnetic field is applied the resulting magnetization is the largest possible for that specific material and size. Here comes the superparamagnetic behaviour, which shares with ferromagnetism the high levels of magnetization reached under the influence of a low magnetic field and with paramagnetism the absence of magnetization when the external magnetic field is removed. The absence enhances the effectiveness of the magnetic separation of cells or biomolecules 1.
Bead Size
We offer magnetic beads in different size, which has an impact on handling and purification results.
Small beads (1-10 µm) : Our supplier magtivio provides beads in three sizes, 600 nm, 1 µm and 3 µm. Advantage are high surface areas available for purification, but a smaller magnetic field, which can negatively influence separation, especially from viscous solutions. In comparison the 600 nm MagSi beads sediment 4x slower than 1.0 µm beads. This allows longer incubation times without shaking, and may be important in automated and other high-throughput applications in which shaking options are often missing. MagSi beads with a diameter of 3 µm have stronger magnetic properties and will separate approximately 4x faster than 600 nm beads under same conditions.
Medium beads (20-40 µm) : Provide efficient separation and large surface area. When combined with agarose surfaces, binding capacities can be in the same range as for small polyvinyl beads. ConA beads of this size, eg ABIN6952467 are an excellent tool in the CUT&RUN and CUT&TAG workflow. Cube Biotech offers MagBeads in the Size of 25 µm; they are used for purification and specific binding of proteins with different tags like GST or Rho1D4.
Large beads (70-120 µm) : have advantages for special applications, e.g. when purification methods contain both magnetic separation and filtration. When combined with agarose surfaces, binding capacities are still high. Examples are the Protein A/G Magnetic Beads or MagSi-S 600 beads by magtivio. 600 nm beads have the advantage of having a larger surface area and the sedimentation time of 600 nm MagSi beads is approximately 4 times slower than that of 1.0 µm beads. This allows longer incubation times without shaking/mixing, and may be important in automated and other high-throughput applications in which shaking/mixing options are often lacking.
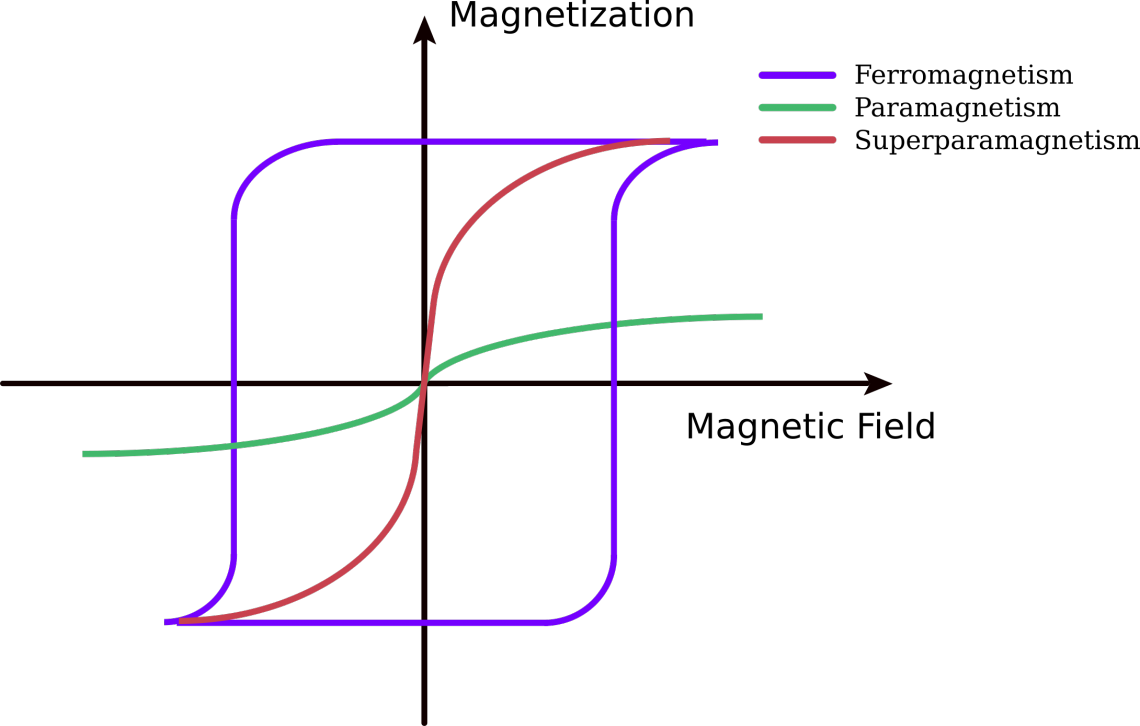
Figure 2. Under the influence of a magnetic field paramagnetic materials are magnetized, but when the magnetic field is removed this magnetization goes to zero. On the contrary, ferromagnetic materials present a remanent magnetization (MR) in the absence of the magnetic field. Superparamagnetic materials share properties of ferromagnetism and paramagnetism.
Discover our Beads & Resin
- Compare and order our bead-based products!
Surface Chemistry
The magnetic core can be covered in a range of different materials, providing different properties.
Agarose forms a three-dimensional hydrophilic mesh out of linear sugar polymers with neutral charge. The Polymer is chemically crosslinked to provide thermal and mechanical stability (see Sepharose). Similar to non-magnetic agarose, it is very well suited to bind to proteins, e.g. via affinity ligands (Tab.1). The large interacting surface leads to high binding capacities. The neutral surface reduces non-specific binding. Cube Biotech and Biovision use this kind of surface in the majority of their products.
Silica beads are most widely used for nucleic acid purification. They become negatively charged at pH >3, and sometimes show unspecific binding in protein purification. magtivio usually works with this surface.
Overview of Coating Materials
| Tag | Description | Protein binding |
|---|---|---|
| Aldehyde | Amine Groups | |
| Alkyl | Typical for reversed phase applications: | Proteins from the following samples: |
| C4-Alkyl | Larger biomolecules like proteins | Cell lysates, culture supernatant (e.g. secreted proteins). |
| C8-Alkyl | suitable for sample preparation for proteomic profiling and biomarker research | Urine, saliva and CSF |
| C18-Alkyl | Peptides and protein digests | Serum and plasma |
| Carboxyl | Couples most proteins | Amine groups; Lysin Histidin etc. |
| ConA | specifically binds to various sugars, glycoproteins, and glycolipids | carbohydrate-binding protein |
| Epoxy | Useful for enzymes | Lysin, Histidin, Tyrosine, etc. |
| GST | Often chosen to promote recombinant protein folding | GST-tagged proteins |
| Hydrazide | Uses aldehyde coupling | Abs and Glycoproteins |
| rho1D4 | 9 aa tag efficient in the purification of membrane proteins | Glycoproteins |
| Streptavidin | 8 aa tag | Biotinylated Protein |
| Tosyl | Useful for antibodies | Sulfhydryl Amine groups |
If u have any further questions concerning beads and their usage feel free to contact our customer support, either via phone live-chat or mail. They will gladly assist you in the right product choice you need.
References:
- : "Nonequilibrium Dynamics of Magnetic Nanoparticles with Applications in Biomedicine." in: Advanced materials (Deerfield Beach, Fla.), Vol. 33, Issue 23, pp. e1904131, (2021) (PubMed).
- : "Magnetic nanoparticles in biomedicine: synthesis, functionalization and applications." in: Nanomedicine (London, England), Vol. 5, Issue 9, pp. 1401-14, (2011) (PubMed).
- : "Magnetic nanoparticles: surface effects and properties related to biomedicine applications." in: International journal of molecular sciences, Vol. 14, Issue 11, pp. 21266-305, (2014) (PubMed).

Creative mind of antibodies-online with a keen eye for details. Proficient in the field of life-science with a passion for plant biotechnology and clinical study design. Responsible for illustrated and written content at antibodies-online as well as supervision of the antibodies-online scholarship program.
Go to author page



