SARS-CoV-2 N-Protein IgG Antibody ELISA Kit
Quick Overview for SARS-CoV-2 N-Protein IgG Antibody ELISA Kit (ABIN6952772)
Target
Reactivity
Detection Method
Method Type
Detection Range
Application
Sample Type
-
-
Minimum Detection Limit
- 3.9 ng/mL
-
Purpose
- For quantitative detection of Anti-SARS-CoV2(N) IgG in serum and plasma.
-
Analytical Method
- Quantitative
-
Sensitivity
- 2.34 ng/mL
-
Components
-
- ELISA Microplate(Dismountable)
- Standard
- Sample/Standard Dilution Buffer
- HRP-labeled Antibody(Concentrated)
- Antibody Dilution Buffer
- TMB Substrate
- Stop Solution
- Wash Buffer(25X)
- Plate Sealer
- Product Description
-
Material not included
-
- Microplate reader (wavelength: 450nm)
- 37 °C incubator
- Automated plate washer
- Precision single and multi-channel pipette and disposable tips
- Clean tubes and Eppendorf tubes
- Deionized or distilled water
-
-
-
-
Comment
-
Washing
Manual: Discard the solution in the plate without touching the side walls. Clap the plate on absorbent filter papers or other absorbent material. Fill each well completely with 350ul wash buffer and soak for 1 to 2 minutes, then aspirate contents from the plate, and clap the plate on absorbent filter papers or other absorbent material.
Automatic: Aspirate all wells, and then wash plate with 350ul wash buffer. After the final wash, invert plate, and clap the plate on absorbent filter papers or other absorbent material. It is recommended that the washer shall be set for soaking 1 minute. (Note: set the height of the needles; be sure the fluid can be sipped up completely) -
Plate
- Pre-coated
-
Protocol
- This kit was based on indirect enzyme-linked immune-sorbent assay technology. Antigen was pre-coated onto 96-well plates. And the HRP conjugated antibody was used as detection antibodies. The standards, test samples and HRP conjugated detection antibody were added to the wells subsequently, and washed with wash buffer. TMB substrates were used to visualize HRP enzymatic reaction. TMB was catalyzed by HRP to produce a blue color product that changed into yellow after adding acidic stop solution. The density of yellow is proportional to the target amount of sample captured in plate. Read the O.D. absorbance at 450nm in a microplate reader, and then the concentration of target can be calculated.
-
Reagent Preparation
-
Wash Buffer Preparation:
If crystals have formed in the concentrate, you can warm it with 40°C water bath (Heating temperature should not exceed 50°C) and mix it gently until the crystals have completely been dissolved. The solution should be cooled to room temperature before use.
Dilute 30ml Concentrated Wash Buffer into 750ml Wash Buffer with deionized or distilled water. Put unused solution back at 2-8°C.
Preparation of HRP-conjugated anti-human IgG Working Solution:
Prepare it within 1 hour before experiment.- Calculate required total volume of the working solution: 50ul / well × quantity of wells. (Allow 55-60ul more than the total volume.)
- Dilute the HRP-conjugated anti-human IgG with Antibody Dilution Buffer at 1:100 and mix them thoroughly. (i.e. Add 1μl HRP-conjugated anti-human IgG into 99μl Antibody Dilution Buffer.)
-
Sample Collection
-
- Serum: Place whole blood sample at room temperature for 2 hours or put it at 2-8°C overnight and centrifugation for 20 minutes at approximately 1000×g, Collect the supernatant and carry out the assay immediately. Blood collection tubes should be disposable, non-pyrogenic, and non-endotoxin.
- Plasma: Collect plasma using (EDTA-Na2 or heparin as an anticoagulant. Centrifuge samples for 15 minutes at 1000×g at 2 - 8°C within 30 minutes of collection. Collect the supernatant and carry out the assay immediately. Avoid hemolysis, high cholesterol samples.
- Other Biological Fluids: Centrifuge samples for 20 minutes at 1000×g at 2-8°C. Collect supernatant and carry out the assay immediately.
-
Sample Preparation
-
The user should estimate the concentration of target protein in the test sample, and select a proper dilution factor to make the diluted target protein concentration fall in the optimal detection range of the kit. Dilute the sample with the provided dilution buffer, and several trials may be necessary. The test sample must be well mi xed with the dilution buffer. And also standard curves and sample should be making in pre-experiment. If samples with very high concentrations, dilute samples with PBS first and then dilute the samples with Sample Dilution.
-
Assay Procedure
-
When diluting samples and reagents, they must be mixed completely and evenly. Before adding TMB into wells, equilibrate TMB Substrate for 30 min at 37 °C. It is recommended to plot a standard curve for each test.
- Set standard, test samples (diluted at least 1/50 with Sample Dilution Buffer), control (blank) wells on the pre-coated plate respectively, and then, records their positions. It is recommended to measure each standard and sample in duplicate. Wash plate 2 times before adding standard, sample and control (blank) wells!
- Prepare Standards: Aliquot 50μl of zero tube, 1sttube, 2ndtube, 3rdtube, 4thtube, 5thtube, 6thtube and Sample Dilution Buffer (blank) into the standard wells.
- Add Samples: Add 50μl of properly diluted sample into test sample wells.
- Incubate: Seal the plate with a cover and incubate at 37°C for 30 minutes.
- Wash: Remove the cover and discard the plate content, and wash plate 3 times with Wash Buffer. Do NOT let the wells dry completely at any time.
- HRP-labeled Antibody: Add 50μl HRP-labeled antibody working solution into above wells (standard, test sample and blank wells). Add the solution at the bottom of each well without touching the sidewall, cover the plate and incubate at 37°C for 30 minutes.
- Wash: Remove the cover, and wash plate 5 times with Wash Buffer, and let the Wash Buffer stay in the wells for 1-2 minute each time.
- TMB Substrate: Add 50μl TMB Substrate into each well, cover the plate and incubate at 37°C in dark within 10-15 minutes. (Note: The reaction time can be shortened or extended according to the actual color change, but not more than 30minutes. You can terminate the reaction when apparent gradient appeared in standard wells.)
- Stop: Add 50μl Stop Solution into each well. The color will turn yellow immediately. The adding order of Stop Solution should be as the same as the TMB Substrate Solution.
- OD Measurement: Read the O.D. absorbance at 450 nm in Microplate Reader immediately after adding the stop solution.
-
Calculation of Results
-
Regarding calculation, (the relative O.D.450) = (the O.D.450 of each well) – (the O.D.450 of blank well). The standard curve can be plotted as the relative O.D.450 of each standard solution (Y) vs. the respective concentration of the standard solution (X). The target concentration of the samples can be interpolated from the standard curve. It is recommended to use some professional software to do this calculation.
-
Restrictions
- For Research Use only
-
-
- by
- Viral Diseases and Infections in Immunodeficiencies Research Group, Institute of Biomedicine of Seville (IbiS)
- No.
- #104432
- Date
- 01/24/2022
- Antigen
- Anti-SARS-CoV-2 N protein IgG
- Lot Number
- H4397H011 E
- Method validated
- ELISA
- Positive Control
Serum from SARS-CoV-2 delta or omicron positive patients
- Negative Control
Blank control (sample dilution buffer)
- Notes
Passed. ABIN6952772 was used to evaluate the concentration of anti-SARS-CoV-2 N-Protein IgG antibodies in sera of patients with confirmed SARS-CoV-2 B.1.617.2 delta or B.1.1.529 omicron infection.
- Primary Antibody
- Secondary Antibody
- Full Protocol
- Collect serum from patients with confirmed SARS-CoV-2 B.1.617.2 delta or B.1.1.529 omicron infection in vacutainer tubes, centrifuge at for 10 min at 2500 rpm and 20°C. Store at -80°C until use.
- Warm the concentrated Wash Buffer in a 40°C water bath and mix it. Dilute 30 ml of concentrated Wash Buffer in 750 ml of deionized water.
- Prepare the HRP working solution 1 h before use. Dilute 60 µL HRP-conjugated anti-human IgG into 5.94 mL Antibody Dilution Buffer.
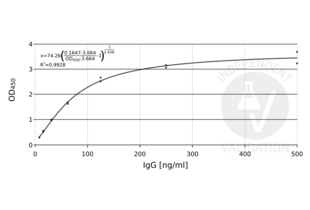
- Prepare a 1:2 dilution series in Sample Dilution Buffer standard curve beginning with 500 ng/mL and consisting of 7 concentrations in total.
- Wash the plate twice with Wash Buffer.
- Add 50 µL of each of the diluted standards, test samples diluted 1:50 in Sample Dilution Buffer, and the blank control in duplicate into the pre-coated plate.
- Cover the plate and incubate for 30 min at 37°C.
- Remove the cover and wash the plate 3 times, making sure the wells don’t be dry completely at any time.
- Add 50 uL of HRP-labeled antibody working solution into each well. Cover the plate and incubate for 30 min at 37°C.
- Wash the plate 5 times, leaving the Wash Buffer a couple of minutes each time.
- Add 50ul TMB Substrate into each well, cover the plate and incubate in the dark for 15 min at 37°C.
- Add 50ul Stop Solution into each well.
- Read absorbance at 450 nm.
- Subtract the mean blank value from the standard and sample measurements. Generate a 4PL standard curve to using GraphPad Prism.
- Experimental Notes
Validation #104432 (ELISA)![Successfully validated 'Independent Validation' Badge]()
![Successfully validated 'Independent Validation' Badge]() Validation ImagesFull Methods
Validation ImagesFull Methods -
-
Storage
- 4 °C
-
Storage Comment
- 2-8°C for 6 months
-
Expiry Date
- 6 months
-
-
-
: "Protein A-Nanoluciferase fusion protein for generalized, sensitive detection of immunoglobulin G." in: Analytical biochemistry, Vol. 660, pp. 114929, (2022) (PubMed).
-
-
- SARS-CoV-2 N-Protein IgG Antibody
-
Target Type
- Antibody
Target
-


 (1 reference)
(1 reference) (1 validation)
(1 validation)



