RFP antibody
-
- Key Features
-
- Frequently cited in scientific publications with 300+ citations
- Validated inhouse for relevant applications
- Extensive validation report for IF provided by one of your peers
- Designed to detect RFP and its variants
- Target See all RFP Antibodies
- RFP (Red Fluorescent Protein (RFP))
- Reactivity
- Discosoma
-
Host
- Rabbit
-
Clonality
- Polyclonal
-
Conjugate
- This RFP antibody is un-conjugated
-
Application
- Western Blotting (WB), ELISA, Immunofluorescence (IF), Immunohistochemistry (IHC), Immunoprecipitation (IP), Flow Cytometry (FACS), Immunohistochemistry (Frozen Sections) (IHC (fro)), Immunohistochemistry (Paraffin-embedded Sections) (IHC (p))
- Purpose
- Polyclonal RFP antibody is designed to detect RFP and its variants.
- Cross-Reactivity (Details)
-
Expect reactivity against RFP and its variants: mCherry, tdTomato, mBanana, mPlum, mOrange and mStrawberry. Assay by immunoelectrophoresis resulted in a single precipitin arc against anti-Rabbit Serum and purified and partially purified Red Fluorescent Protein (Discosoma).
No reaction was observed against Human, Mouse or Rat serum proteins. - Purification
- This product was prepared from monospecific antiserum by immunoaffinity chromatography using Red Fluorescent Protein (Discosoma) coupled to agarose beads followed by solid phase adsorption(s) to remove any unwanted reactivities.
- Sterility
- Sterile filtered
- Immunogen
-
The immunogen is a Red Fluorescent Protein (RFP) fusion protein corresponding to the full length amino acid sequence (234aa) derived from the mushroom polyp coral Discosoma.
Immunogen Type: Recombinant Protein - Isotype
- IgG
- Product Specific Information
-
What can the RFP antibody ABIN129578 be used for? This polyclonal RFP antibody detects RFP and its variants, such as mCherry, tdTomato, mBanana, mOrange and others. The RFP antibody has been validated for various applications and can be used for the detection of RFP and derivatives by ELISA, immunofluorescence, FACS, immunohistochemistry or western blotting.
What validation data is available for this RFP antibody? The antibody is referenced in several hundred publications and is characterized by a proven, very high reliability. It has currently 12 product images that show its performance in a variety of applications. The product is currently available in 10µl and 100µl quantities. RFP antibody for the detection of Red fluorescent protein and derivatives.
What is the function of RFP? RFP is a fluorophore that fluoresces red-orange when excited. The protein has many applications, including as a marker in monitoring physiological processes in vivo. The excitation maximum is about 555nm, the emission maximum is 584nm. RFP antibodies provide a fairly easy-to-use way to visualize RFP in its various applications.
-
-
- Application Notes
-
Suggested dilutions:
ELISA: 1:28,700 - 1:48,700
IF: 1:200 - 1:2,000
FACS: 1:200 - 1:2,000
WB: 1:1,000 - 1:5,000
IHC: 1:200 - 1:2,000
IP: User Optimized - Comment
-
This RFP antibody can be used to detect RFP by ELISA (sandwich or capture) for the direct binding of antigen.
Optimal titers for applications should be determined by the researcher. - Restrictions
- For Research Use only
-
- by
- Centre d'Immunologie de Marseille-Luminy
- No.
- #101103
- Date
- 03/07/2017
- Antigen
- RFP
- Lot Number
- 34945
- Method validated
- Immunofluorescence
- Positive Control
- CX3CL1mcherry+ murine thymus
- Negative Control
- CX3CL1 WT murine thymus
- Notes
Passed. The RFP antibody ABIN129578 specifically labels RFP+ cells in CX3CL1mcherry+ murine thymus in immunofluorescence, consistent with the expression pattern of CX3CL1mcherry.
- Primary Antibody
- ABIN1043867
- Secondary Antibody
- donkey Fab'2 anti-rabbit A647 conjugated antibody (Jackson Immunoresearch, 711-606-152, lot 128806)
- Full Protocol
- Harvest thymus from 7 weeks old mice in 1X Dulbecco's phosphate buffered saline (DPBS) (Gibco Life Technologies, 14200-067).
- Fix mouse thymus in antigen fix (Diapath, P0014) for 2h at 4?C.
- Wash tissue in 0.1M pH7.4 phosphate buffer at for 1h at 4?C.
- Dehydrate tissue in 30% sucrose solution at 4?C ON.
- Snap freeze tissue in Tissue Freezing Medium (ElectroMicroscopy Science, 72592-C) at -80?C.
- Cut blocks into 25?m sections using a cryostat (Leica, CM3050 S).
- Transfer sections to a slide.
- Create a hydrophobic barrier on the slide around sections with Dako pen (Dako, S2002, lot 00081640).
- Place slide in a humidified chamber and rehydrate sections in 0.1 M TrisHCl pH7.4 for 10min at RT.
- Gently remove buffer by tapping slide.
- Permeabilize tissue in 0.1M TrisHCl pH7.4 containing 2% Triton X-100 (Sigma, lot 015K0039) and 0.5% BSA, for 20min at RT.
- Incubate sections with primary RFP antibody (Red Fluorescent Protein) (AA 234) (antibodies-online, ABIN129578, lot 34945) diluted 1:1000 0.1M TrisHCl pH7.4 containing 2% Triton X-100 (Sigma, batch 015K0039) and 0.5% BSA for 2h at RT.
- Incubate a no primary antibody negative control in parallel in 0.1M TrisHCl pH7.4 containing 2% Triton X-100 (Sigma, batch 015K0039) and 0.5% BSA.
- Wash slides 1x 5min in 0.1M TrisHCl pH7.4.
- Incubate sections with secondary donkey Fab'2 anti-rabbit AF647 conjugated antibody (Jackson Immunoresearch, 711-606-152, lot 128806) diluted 1:300 in 0.1M TrisHCl pH7.4 containing 2% Triton X-100 (Sigma, batch 015K0039) and 0.5% BSA for 2h at RT.
- Wash slides 1x 5min in 0.1M TrisHCl pH7.4.
- Add approximately 10µL of Slowfade Gold antifade reagent (ThermoFisher Scientific, S36937, lot 1226836) for each section and mount cover slip.
- Image acquisition on an LSM 880 (Zeiss), 20x magnification, 1000 resolution.
- Experimental Notes
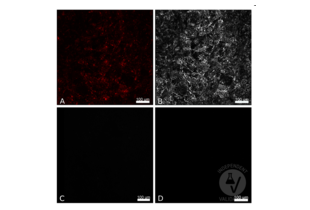
Validation #101103 (Immunofluorescence)![Successfully validated 'Independent Validation' Badge]()
![Successfully validated 'Independent Validation' Badge]() Validation Images
Validation Images![CX3CL1 positive cells in CX3CL1mcherry murine thymus (A, mCherry) were stained with ABIN1043867 and an AF647 conjugated secondary antibody (B) or with the secondary antibody alone (C). CX3CL1 positive cells in CX3CL1 wt murine thymus were stained with ABIN1043867 and the AF647 conjugated antibody (D). Scale bar 100?m, 20x magnification, 1000 resolution]() CX3CL1 positive cells in CX3CL1mcherry murine thymus (A, mCherry) were stained with ABIN1043867 and an AF647 conjugated secondary antibody (B) or with the secondary antibody alone (C). CX3CL1 positive cells in CX3CL1 wt murine thymus were stained with ABIN1043867 and the AF647 conjugated antibody (D). Scale bar 100?m, 20x magnification, 1000 resolution
Full Methods
CX3CL1 positive cells in CX3CL1mcherry murine thymus (A, mCherry) were stained with ABIN1043867 and an AF647 conjugated secondary antibody (B) or with the secondary antibody alone (C). CX3CL1 positive cells in CX3CL1 wt murine thymus were stained with ABIN1043867 and the AF647 conjugated antibody (D). Scale bar 100?m, 20x magnification, 1000 resolution
Full Methods -
- Format
- Liquid
- Concentration
- 1.16 mg/mL
- Buffer
- 0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2, 0.01% (w/v) Sodium Azide
- Preservative
- Sodium azide
- Precaution of Use
- This product contains sodium azide: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
- Handling Advice
- Avoid cycles of freezing and thawing.
- Storage
- 4 °C/-20 °C
- Storage Comment
- Store vial at -20° C prior to opening. Aliquot contents and freeze at -20° C or below for extended storage. Centrifuge product if not completely clear after standing at room temperature. This product is stable for several weeks at 4° C as an undiluted liquid. Dilute only prior to immediate use.
- Expiry Date
- 12 months
-
-
: "Injury primes mutation-bearing astrocytes for dedifferentiation in later life." in: Current biology : CB, (2023) (PubMed).
: "Diet suppresses glioblastoma initiation in mice by maintaining quiescence of mutation-bearing neural stem cells." in: Developmental cell, (2023) (PubMed).
: "Molecular sensing of mechano- and ligand-dependent adhesion GPCR dissociation." in: Nature, Vol. 615, Issue 7954, pp. 945-953, (2023) (PubMed).
: "Overexpression of Lin28A in neural progenitor cells in vivo does not lead to brain tumor formation but results in reduced spine density." in: Acta neuropathologica communications, Vol. 9, Issue 1, pp. 185, (2022) (PubMed).
: "A versatile viral toolkit for functional discovery in the nervous system. ..." in: Cell reports methods, Vol. 2, Issue 6, pp. 100225, (2022) (PubMed).
: "Axon guidance at the spinal cord midline-A live imaging perspective." in: The Journal of comparative neurology, (2021) (PubMed).
: "Extrinsic Regulators of mRNA Translation in Developing Brain: Story of WNTs." in: Cells, Vol. 10, Issue 2, (2021) (PubMed).
: "Co-activation of Sonic hedgehog and Wnt signaling in murine retinal precursor cells drives ocular lesions with features of intraocular medulloepithelioma." in: Oncogenesis, Vol. 10, Issue 11, pp. 78, (2021) (PubMed).
: "The white matter is a pro-differentiative niche for glioblastoma." in: Nature communications, Vol. 12, Issue 1, pp. 2184, (2021) (PubMed).
: "Antinociceptive modulation by the adhesion GPCR CIRL promotes mechanosensory signal discrimination." in: eLife, Vol. 9, (2021) (PubMed).
: "Reticular Fibroblasts Expressing the Transcription Factor WT1 Define a Stromal Niche that Maintains and Replenishes Splenic Red Pulp Macrophages." in: Immunity, Vol. 53, Issue 1, pp. 127-142.e7, (2020) (PubMed).
: "Transcriptional activities of human elongation factor-1α and cytomegalovirus promoter in transgenic dogs generated by somatic cell nuclear transfer." in: PLoS ONE, Vol. 15, Issue 6, pp. e0233784, (2020) (PubMed).
: "Gastric squamous-columnar junction contains a large pool of cancer-prone immature osteopontin responsive Lgr5-CD44+ cells." in: Nature communications, Vol. 11, Issue 1, pp. 84, (2020) (PubMed).
: "Yorkie and JNK revert syncytial muscles into myoblasts during Org-1-dependent lineage reprogramming." in: The Journal of cell biology, Vol. 218, Issue 11, pp. 3572-3582, (2020) (PubMed).
: "Evolution of Ovipositor Length in Drosophila suzukii Is Driven by Enhanced Cell Size Expansion and Anisotropic Tissue Reorganization." in: Current biology : CB, Vol. 29, Issue 12, pp. 2075-2082.e6, (2020) (PubMed).
: "Disynaptic cerebrocerebellar pathways originating from multiple functionally distinct cortical areas." in: eLife, Vol. 9, (2020) (PubMed).
: "Cellular and molecular properties of neural progenitors in the developing mammalian hypothalamus." in: Nature communications, Vol. 11, Issue 1, pp. 4063, (2020) (PubMed).
: "From 2D to 3D: Promising Advances in Imaging Lung Structure." in: Frontiers in medicine, Vol. 7, pp. 343, (2020) (PubMed).
: "Osterix-Cre marks distinct subsets of CD45- and CD45+ stromal populations in extra-skeletal tumors with pro-tumorigenic characteristics." in: eLife, Vol. 9, (2020) (PubMed).
: "Genetic background mutations drive neural circuit hyperconnectivity in a fragile X syndrome model." in: BMC biology, Vol. 18, Issue 1, pp. 94, (2020) (PubMed).
-
: "Injury primes mutation-bearing astrocytes for dedifferentiation in later life." in: Current biology : CB, (2023) (PubMed).
-
- Target
- RFP (Red Fluorescent Protein (RFP))
- Alternative Name
- RFP (RFP Products)
- Background
-
Synonyms: DsRed, rDsRed, Discosoma sp. Red Fluorescent Protein, Red fluorescent protein drFP583
Fluorescent proteins such as Discosoma Red Fluorescent Protein (DsRed) from sea anemone Discosoma sp. mushroom or green fluorescent protein (GFP) from Aequorea victoria jellyfish are widely used in research practice. Fusion RFP and GFP commonly serve as marker for gene expression and protein localization. As DsRed and GFP share only 19% identity, therefore, in general, anti-GFP antibodies do not recognize DsRed protein and vice versa. Structurally, Discosoma red fluorescent protein is similar to Aequorea green fluorescent protein in terms of its overall fold (a β-can) and chromophore-formation chemistry. However, Discosoma red fluorescent protein undergoes an additional step in the chromophore maturation and obligates tetrameric structure.
-

 (356 references)
(356 references) (1 validation)
(1 validation)



