Antibodies are proteins produced in vertebrates in response to certain substances. Antibodies are an important element of the so-called humoral (antibody-mediated) immunity. They belong to the gamma globulin fraction and are also called immunoglobulins (Ig).
Antibodies are produced by the body when B cells come in contact with a matching antigen. As a result, the B cell is activated and differentiates into a plasma cell that secretes large amounts of antibody. These antibodies are able to specifically bind the antigen which has caused the differentiation of the B cell.
Structure
Antibodies have a common basic structure: each consists of two heavy and two light identical polypeptide chains, linked via disulfide bridges. The heavy and light chains include constant and variable regions. The variable regions of both chains determine the binding domain (paratope) for a specific antigenic determinant (epitope). The epitope is the part of the antigen that is recognized by the antibody. In addition, the constant regions of the heavy chains determine the immunoglobulin class to which the antibody belongs ( IgA , IgD , IgE ,
, , ). The immunoglobulin class is also called isotype.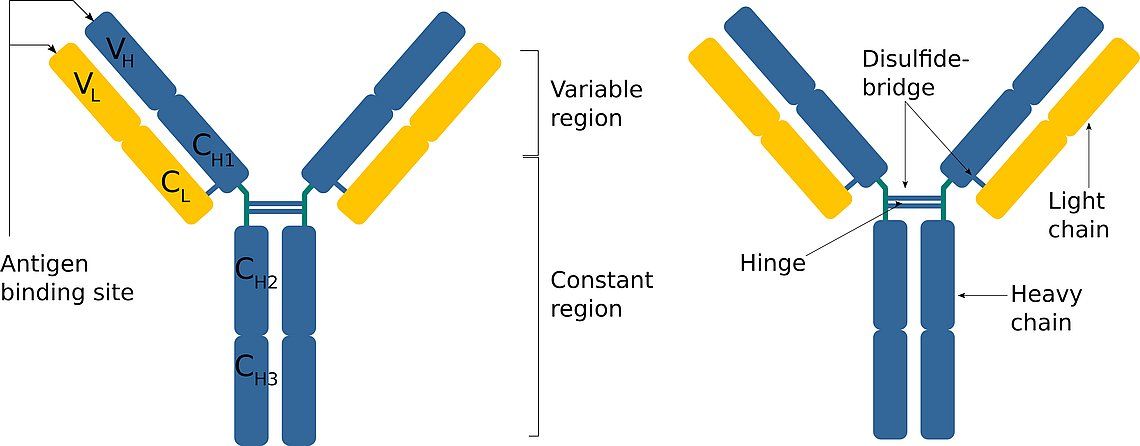
Left: Schematic structure of an IgG antibody. Each antibody molecule consists of two heavy (blue) and two light (yellow) chains, linked by disulfide bridges. These also form the so-called hinge region which connects the two heavy chains. Right: Composition of the heavy and light chainsThe heavy and light chains are composed of individual domains. Both the heavy and the light chains include constant (CL, CH) and variable (VL, VH) domains. The variable domains of both chains determine the specificity of the antigen binding site. The heavy chain constant regions determine which antibody class the antibody belongs to.
The various antibody classes are found in different compartments of the body. For example, IgA is present in the saliva while IgG and IgM are found in the blood. In addition, membrane-bound antibodies are also found (e.g.: IgE on mast cells or IgD on B lymphocytes).
Custom Antibody Services
Custom Antibody Services
Benefit from combined expertise of antibodies-online and our Partner Rockland Immunochemicals, Inc. Collaborative, experienced, and conducted completely in our own facilities, our custom antibody production services can drive your project from the design and synthesis of your antigen through to production and characterization of your antibody.
Benefit from combined expertise of antibodies-online and our Partner Rockland Immunochemicals, Inc. Collaborative, experienced, and conducted completely in our own facilities, our custom antibody production services can drive your project from the design and synthesis of your antigen through to production and characterization of your antibody.
Function
In the course of an immune response, the antibodies perform various functions. They capture and block invading antigens so that they cannot exert their harmful effect, or they prevent the antigen’s interaction with body cells (e.g. to prevent the penetration of viruses and bacteria into body cells).
Antibodies also opsonize pathogens. In this case the antibody constitutes a marker, e.g. for phagocytes. The phagocytes interact with the constant regions of the antibody in contact and are induced to uptake and digest the pathogen (e.g., a bacterium).
Antibodies may also bind to body cells, thereby causing the so-called NK cells (natural killer cells) to selectively kill the cell. This is useful when a cell is infected with a pathogen (e.g., a virus). Other important functions include the activation of various cell types of the immune system and the complement system.
Application
Antibodies are used in medicine for diagnostic and therapeutic purposes (e.g. serological tests, diagnosis and follow-up of infectious diseases, passive immunization, etc.). They also offer many possible uses (ELISA, FACS, Western blots, immunohistochemistry, etc.) for research purposes. For these purposes, polyclonal antibodies, monoclonal antibodies or recombinant antibodies are used.
Features Antibodies
- (363)
- (11)
- (1)
- (272)
- (43)
- (63)
- (41)
- (2)
- (14)
- (12)
- (1)
- (11)
- (14)
- (1)
- (12)
- (15)
- (1)
- (15)
- (17)
- (1)
- (15)
- (5)
- (1)
- (21)
- (6)
- (1)
- (19)
- (7)
- (1)

Master of Science in engineering. 12+ years of experience in marketing and e-commerce in the life science sector.
Go to author page



