Smartox Biotechnology: Synthetic Polypeptide Toxins for Research Applications
Written/Edited by Dr. Ryan Robinson, PhDToxins are broadly defined as bioactive products, produced in a living organisms, which are capable of inducing a disease state upon contact or absorption. Succinctly, a toxin may be described as a biologically produced poisonous substance. Toxins vary significantly with respect to size, structure, and mechanism-of-action. They range from small, simple, organic molecules like bacterial lipopolysaccharides (LPS) to larger more complex polypeptide structures like apamin and conotoxins.
antibodies-online offers more than 70 synthetically-produced polypeptide toxins from leading manufacturer Smartox Biotechnology. Smartox Biotechnology is an industry pioneer in the development and production of animal toxins for biological research, offering research-grade toxin compounds that allow for specific targeting and inhibition of cell-surface receptors and ion-channels.
Toxins have always captured the interest of curious biomedical and life-science researchers. Early toxicological research predominantly focused on development of therapeutic countermeasures and understanding of the evolutionary framework under which biological toxin development originated.
Toxins Are Diverse in Structure and Function, They Commonly Effect Physiology By:
| Blocking cell surface receptors and ion channels. | Iberiotoxin from the East Indian red scorpion, Buthus tamulus selectively binds and inhibits ion channels, blocking cell physiological function. 1 |
| Chemically or enzymatically destroying cells. | Haemotoxic enzymes, present in the venom of some species of snakes and spiders, will target and destroy erythrocytes and platelets. 2 |
| Stimulating an overwhelming and damaging immune response. | Lipopolysaccharide (or LPS) is a bacterial endotoxin and a core component of the outer membrane in Gram-negative bacteria. LPS release from bacterial infection can lead to a massive, overwhelming immune response and development of septic shock. 3 |
Toxins have always captured the interest of curious biomedical and life-science researchers. Early toxicological research predominantly focused on development of therapeutic countermeasures and understanding of the evolutionary framework under which biological toxin development originated. As toxicology research progressed, the increased understanding of toxin structure and function provided researchers in other fields an opportunity to utilize naturally produced toxins as tools to study other relevant biological interests.
Polypeptide Toxins Are Potent, Specific Ion-Channel Inhibitors
Ion-channels and cell-surface receptors are highly specialized protein complexes that mediate communication between an individual cell and the environment. These structures are highly conserved, and ubiquitously expressed in multicellular life, but are found at particularly high density dense on highly communicative cells within the immune and nervous systems. In these systems, ion-channels and cell surface receptors mediate intercellular communication, and modulate the cell’s detection of and response to its environment.
Loss-of-function experiments are a very common method for studying the function of individual ion-channels and cell-surface receptors. In these experiments a researcher limits or eliminates the function of a target structure and observes the result.
Loss-of-function experiments demand both a high degree of specificity, and a robust inhibition of the target studied. Historically, they have relied upon labor-intensive genetic techniques designed to disrupt expression of the gene coding for the target of interest, or the use of “blocking antibodies” which have a longstanding reputation for limited efficacy.
For many targets, application of highly specific toxin is a powerful alternative approach. Application of a polypeptide toxin is simpler than generation of a knockout animal. Toxins also supersede the efficacy of most blocking antibodies by virtue of their intrinsic functionality as blocking reagents. Generally speaking, the precise, evolutionarily derived function of a toxin is to recognize and disrupt physiology by blocking the activity of its target. By contrast the antibody principally evolved to elicit a systemic immune response against a specific target, and displays a less robust disruption of target function.
Blocking Antibodies
Comparably low target specificity
Generally recognize linear epitopes that may be masked on a native target
Mediocre at blocking native protein functional activity
Toxins

Exceptionally high target specificity
Recognize native three-dimensional conformational structures
Exceptional at blocking native protein functionality
Toxins also possess other unique attributes that allow them to outperform antibodies for certain specific functions. Small size, minimal complexity, and high stability permit toxin use in applications and scenarios where use of an antibody is nontrivial.
The potency, specificity, and unique physical properties possessed by many toxins makes them laudable blocking reagents for studying cellular function and physiology. In fact, toxins like Stichodactyla Toxin (ShK) are potent enough to recognize, bind, and inhibit voltage gated potassium channels at trace concentrations (picomolar to nanomolar) while simultaneously avoiding disruption of normal physiological function in other similar ion-channels.
Toxins: Versatile, Simple, Reliable Reagents for Interdisciplinary Research
Historically, toxins have been most commonly used in neuroscience research. To study neurophysiology researchers have traditionally applied various neuroactive toxic compounds to disrupt ion-channel function. Use of toxins in this manner has significantly advanced understanding of the nervous system, and the ability to block activity of individual ion-channel species has permitted systematic characterization of nervous system function at the sub-cellular level.
While neuroscience and neurophysiology research represent be the canonical center of toxin research use, the diversity in toxin structure and function makes them invaluable tools for studying cellular function across many different cell types, and in countless research fields.
Learn How Different Toxins Are Used in Various Research Disciplines:
Iberiotoxin
- Iberiotoxin is a 37-AA polypeptide derived from the eastern Indian red scorpion Buthus tamulus.
- Iberiotoxin binds and selectively inhibits calcium activated potassium channels, blocking neurophysiological function.
- Iberiotoxin is commonly used for researching smooth muscle physiology, including vascular and contraction and dilation.
- Selected References: PMC2992907, PMID23861849
APETx2
- APETx2 is a 42-aa polypeptide toxin derived from the sea anemone Anthopleura elegantissima.
- APETx2 binds and inhibits many acid sensing ion channels.
- APETx2 is particularly useful for researching sensory and pain conduction and cytokine mediated inflammation.
- Selected References: PMC2992907, PMID25687542 , PMID25073401
ProTX-II
- ProToxin-II is a 30-aa polypeptide toxin isolated from the Peruvian green velvet tarantula Thrixopelma pruriens.
- ProToxin-II selectively binds and inhibits voltage gated sodium channels. ProToxin-II provides more robust inhibition than Tetrodotoxin, inhibiting both TTX-sensitive and TTX-resistant ion channels.
- ProToxin-II is useful for researching pain sensation, conduction, and management. The therapeutic potential of ProToxin-II for pain management is currently being investigated.
- Selected References: PMID12475222, PMID25026046, PMID25818052
Ω-conotoxin / GVIA
- Omega-conotoxin GVIA is a 27-aa polypeptide toxin of the conotoxin family, it was originally isolated from the cone snail Conus geographus.
- Omega-conotoxin GVIA selectively binds and inhibits a single voltage gaged calcium ion cahnnel, Ca+2.2
- Omega-conotoxin GVIA is used by researchers studying synaptic transmission, sensation, neurological function, and neurological degeneration.
- Selected References: PMID2845019, PMID25195871
GsMTx4
- GsMTx4 is a 34-AA polypeptide toxin originally isolated from the Chilean rose tarantula Grammostola rosea.
- GsMTx4 perturbs the natural connection between mechanosensitive ion channels and the lipid membrane. GsMTx4 is able to block activation of these ion channels without having to physically interact with or bind them.
- GsMTx4 is used to research mechanonsensation, cardiovascular disease, and mechanonsensory neuron development.
- Selected References: PMID21696149, PMID25938516, PMID24306105
ShK
- Stichodactyla Toxin is a 35-AA polypeptide toxin originally isolated from the Caribbean sea anemone Stoichactis helianthus.
- Stichodactyla Toxin binds and inhibits voltage gated potassium channels , , , .
- Stichodactyla Toxin is frequently used to research immune response, autoimmunity, inflammation, and response to injury.
- Selected References: PMID24891519, PMID24305723, PMID21824083
- The high affinity and specificity displayed by stichodactyla toxin have allowed it to be utilized in particularly novel ways. Smartox biotechnology product ABIN2566196 (TMR-ShK) is comprised of a synthetic stichodactyla toxin conjugated to a bright red fluorescent probe (TMR). This makes the the toxin useful for labeling voltage gated potassium channels in IF and flow cytometry experiments. Read more about TMR-ShK below.
Pertussis Toxin
- Pertussis toxin is multi-subunit protein toxin isolated from the bacterium Bordetella pertussis, the infectious agent responsible for whooping cough.
- Pertussis toxin blocks interaction between G proteins and G protein coupled receptors. This blocks intracellular communication, particularly in cells responsible for innate immunity
- Pertussis Toxin is frequently used to research innate immune response, immune cell activation, chemotaxis, and chemokine production.
- Selected References: PMID2351723
Genetically-Engineered Synthetic Toxins Reveal a Wealth of Research Opportunities
For decades, researchers have utilized toxins to strategically probe the role that different ion channels play in synaptic transmission, neural function, and physiology. Traditionally, toxin use in neuroscience research has been restricted by the natural properties of the toxin used, and by extension its ability to augment ion-channel function.
Recently, researchers have combined advances in genetic engineering with an in-depth knowledge of toxin structure to develop specifically engineered toxins that supersede the abilities of their natural precursors.
For example, product ABIN2566196 from Smartox biotechnology is a synthetically produced Stichodactyla toxin fusion protein that includes a bright red fluorophore (TMR). Stichodactyla toxin has an exceptionally high affinity for the voltage gated potassium channel KCNA3/Kv1.3, and product ABIN2566196 is able to fluorescently label this ion channel. ABIN2566196 may be used in place of an antibody for flow cytometry and immunofluorescence experiments.
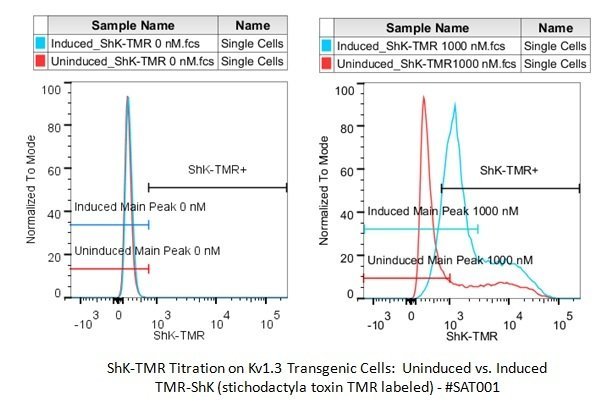
Fluorescence intensity in the red channel is measured by FACS for induced transgenic cells overexpressing Kv1.3 (blue histogram) and an uninduced control population (red histogram).
(Left)Negative control - No fluorescent labeling for either population
(Right)Both populations labeled with 1000nm ABIN2566196 . The uninduced control population (blue histogram) displays no appreciable staining, while the induced population overexpressing Kv1.3 (red histogram) is clearly labeled with strong fluorescent signal.
The ability to accurately label Kv1.3 with a high affinity fluorescent probe has broad research implications that extend beyond the ion-channel’s canonical role in neurophysiology. For example, upregulation of Kv1.3 expression in autoreactive T-cells has been implicated in several autoimmune disorders, including psoriasis and ulcerative colitis4. Using ABIN2566196 a researcher can simply and effectively label Kv1.3 in T-cell populations, permitting rapid, high-throughput analysis of autoreactive T-cell development through flow-cytometry or other fluorescent detection methods5.
Niche Roles, Once Filled by Antibodies, May Soon Be Occupied by Synthetically Produced Toxins
The narrative surrounding the study, development, use, and optimization of toxins for life-science and biomedical research bares a striking resemblance to a similar story of another common reagent, the antibody. Both antibodies and toxins are explicitly natural in origin. Their common laboratory use was born from curiosity and intense study of observed phenomena in a particular subset of species, and both were eventually co-opted by industrious scientists who sought an opportunity to utilize them for purposes beyond their natural, biological role.
Today, the once humble antibody is widely recognized as the canonical proteomics research reagent. To claim that antibody use is ubiquitous would be an understatement; antibodies are utilized in nearly every life-science research laboratory across the globe, conjugated to an innumerable quantity of detection conjugates, and reactive against hundreds of thousands of specific targets derived from nearly any species imaginable.
However, for all its prolificity, the antibody does have a few well-established limitations. These have become an increasingly salient concern as new targets and novel methods have captivated the interest of eager life-science researchers. For now, the antibody’s role in research is set. However, the steady march of progress has driven researchers to adopt an ever-increasing panoply of alternative options, which are frequently less expensive, simpler, and more efficient.
As the sun sets on the age of the antibody, natural and synthetically reproduced toxins will inevitably fill some of the roles once occupied by these academic and industry titans of the laboratory. The diverse structure of various peptide toxins, and their unique capability to specifically and potently inhibit ion-channel functionality makes them supremely useful for studying physiology at the cellular level. Use of specific toxins in conjunction with fluorescent substrates to label targeted cell-surface receptors in a highly specific manner further highlights the diverse research possibilities possessed by this unique class of molecules.
Indeed, as the use of research antibodies becomes progressively eclipsed by alternative options, and as advanced genetic engineering techniques allow for more rapid production, modulation, and optimization of synthetic toxin products, we will likely find ourselves in the wake of a toxin research revolution.
For more information on using toxins in biological research, or to investigate a specific product, please contact antibodies-online scientific support: support@antibodies-online.com.

Offered pre- and post-sales consultation and support to antibodies-online customers. Provided troubleshooting and technical assistance with proteomics and genomics experiments. Composed and distributed cross-channel digital marketing campaigns.
Go to author page



