Vitronectin Antibodies
Your search for reliable Vitronectin antibodies ends here. Vitronectin, known by aliases such as VTN, vtn, vtna, vtnb, CpipJ_CPIJ016691, vtn.L, Vtn, is an integral part of our antibody range. Whether you're working with Human, Mouse, Rat, Cow, Dog, or other species, our range of Vitronectin antibodies offer precise detection across diverse samples. These specialized antibodies are tailored for various scientific applications like WB, ELISA, IHC, FACS, IF (cc), providing you with options like polyclonal, recombinant, and monoclonal antibodies, sourced from different host species such as Rabbit, Mouse, Sheep. The efficacy of our antibodies is well-established, demonstrated through multiple methods.
Detailed information, including references, images, and validations by other customers, can be found on each product page. Should you require assistance in finding a specific product, our customer service team is ready to assist. Utilize our Vitronectin antibodies in your research endeavors for dependable Vitronectin detection.
VTN Reactivity: Human ELISA, IHC, FACS Host: Mouse Monoclonal 1G11E8 unconjugated

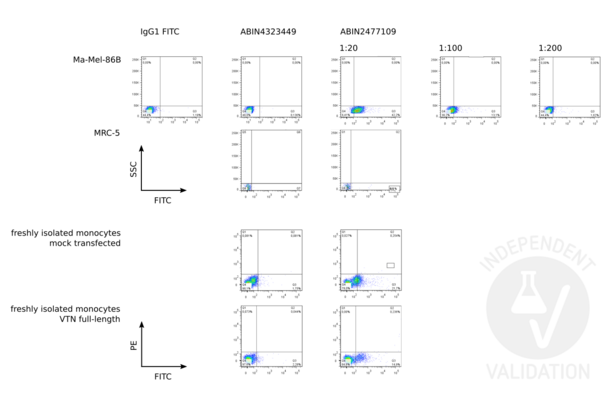 Vitronectin antibody (ABIN2477109)
Vitronectin antibody (ABIN2477109)
VTN Reactivity: Human WB, ELISA, IHC (fro) Host: Mouse Monoclonal 8E6 (1-110) unconjugated
VTN Reactivity: Human WB, ELISA, IHC, IP Host: Rabbit Polyclonal unconjugated
Vitronectin Antibodies by Reactivity
Find Vitronectin Antibodies for a variety of species such as anti-Human Vitronectin, anti-Mouse Vitronectin, anti-Rat Vitronectin. The species listed below are among those available. Click on a link to go to the corresponding products.
Vitronectin Antibodies by Host
Find Vitronectin Antibodies with a specific Host. The Host listed below are among those available. Click on a link to go to the corresponding products.
Vitronectin Antibodies by Clonality
Find available monoclonal or polyclonal Vitronectin Antibodies. Click on a link to go to the corresponding products.
Popular Vitronectin Antibodies
- (6)
- (1)
- (4)
- (4)
- (3)
- (3)
- (3)
- (2)
- (2)
- (2)
- (3)
- (3)
- (1)
- (2)
- (3)
- (1)
Latest Publications for our Vitronectin Antibodies
: "Structure of human Vitronectin C-terminal domain and interaction with Yersinia pestis outer membrane protein Ail." in: Science advances, Vol. 5, Issue 9, pp. eaax5068, (2020) (PubMed).: "Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury." in: Journal of biomedical materials research. Part A, Vol. 104, Issue 8, pp. 1902-11, (2017) (PubMed).
: "Selection of a Novel Aptamer Against Vitronectin Using Capillary Electrophoresis and Next Generation Sequencing." in: Molecular therapy. Nucleic acids, Vol. 5, Issue 11, pp. e386, (2016) (PubMed).
: "Effect of Puumala hantavirus infection on human umbilical vein endothelial cell hemostatic function: platelet interactions, increased tissue factor expression and fibrinolysis regulator release." in: Frontiers in microbiology, Vol. 6, pp. 220, (2015) (PubMed).
: "Losartan ameliorates dystrophic epidermolysis bullosa and uncovers new disease mechanisms." in: EMBO molecular medicine, Vol. 7, Issue 9, pp. 1211-28, (2015) (PubMed).
: "Induced apoptosis of osteoblasts proliferating on polyhydroxyalkanoates." in: Biomaterials, Vol. 34, Issue 15, pp. 3737-46, (2013) (PubMed).
: "The Hippo pathway target, YAP, promotes metastasis through its TEAD-interaction domain." in: Proceedings of the National Academy of Sciences of the United States of America, Vol. 109, Issue 37, pp. E2441-50, (2012) (PubMed).
: "A proteomics approach to identify changes in protein profiles in serum of Familial Adenomatous Polyposis patients." in: Cancer letters, Vol. 272, Issue 1, pp. 40-52, (2009) (PubMed).
: "The contributions of integrin affinity and integrin-cytoskeletal engagement in endothelial and smooth muscle cell adhesion to vitronectin." in: The Journal of biological chemistry, Vol. 282, Issue 21, pp. 15679-89, (2007) (PubMed).
: "Vitronectin in atherosclerotic disease." in: Clinica chimica acta; international journal of clinical chemistry, Vol. 368, Issue 1-2, pp. 77-83, (2006) (PubMed).
Aliases for Vitronectin Antibodies
vitronectin (VTN) Antibodiesvitronectin (vtn) Antibodies
vitronectin a (vtna) Antibodies
vitronectin b (vtnb) Antibodies
vitronectin (CpipJ_CPIJ016691) Antibodies
vitronectin L homeolog (vtn.L) Antibodies
vitronectin (Vtn) Antibodies
Aa1018 Antibodies
AI256434 Antibodies
DKFZp470F0511 Antibodies
LOC100228142 Antibodies
V75 Antibodies
VN Antibodies
Vn Antibodies
VNT Antibodies
VTN Antibodies
vtn Antibodies
zgc:112277 Antibodies
Did you look for something else?
- Vitrin Antibodies
- Vitellogenin Antibodies
- Vitamin E Antibodies
- Vitamin D3 Antibodies
- Vitamin D-Binding Protein Antibodies
- Vitamin D Receptor Antibodies
- Vitamin B6 Antibodies
- Vitamin B2 Antibodies
- Vitamin B12 Antibodies
- Visual System Homeobox 2 Antibodies
- VISTA Antibodies
- VIRMA/KIAA1429 Antibodies
- VIPR2 Antibodies
- VIPR1 Antibodies
- VIPAR Antibodies
- Vip Antibodies
- Vinexin Antibodies
- Vinculin Antibodies
- Vimentin Antibodies
- Villin 1 Antibodies
- VKORC1 Antibodies
- VKORC1L1 Antibodies
- VLDLR Antibodies
- VMA21 Antibodies
- VMO1 Antibodies
- VMP1 Antibodies
- VN1R1 Antibodies
- VN1R2 Antibodies
- VN1R4 Antibodies
- VNN1 Antibodies
- VNN2 Antibodies
- VNN3 Antibodies
- Vomeronasal 1 Receptor 3 Antibodies
- Vomeronasal 1 Receptor 5 Antibodies
- VOPP1 Antibodies
- VPRBP Antibodies
- VPREB1 Antibodies
- VPREB3 Antibodies
- VPS11 Antibodies
- VPS13A Antibodies




