IL-8 ELISA Kit
Quick Overview for IL-8 ELISA Kit (ABIN414887)
Target
See all IL-8 (IL8) ELISA KitsReactivity
Detection Method
Method Type
Detection Range
Application
Sample Type
-
-
Minimum Detection Limit
- 15.6 pg/mL
-
Purpose
-
The kit is a sandwich enzyme immunoassay for in vitro quantitative measurement of IL8 in human serum, plasma, tissue homogenates, cell lysates, cell culture supernates.
We offer validation data (WB) for the kit components. So you can be sure to order a reliable ELISA kit product composed of high quality reagents. -
Analytical Method
- Quantitative
-
Specificity
-
This assay has high sensitivity and excellent specificity for detection of Interleukin 8 (IL8).
No significant cross-reactivity or interference between Interleukin 8 (IL8) and analogues was observed. -
Cross-Reactivity (Details)
- No significant cross-reactivity or interference between Interleukin 8 (IL8) and analogues was observed.
-
Sensitivity
- 5.7 pg/mL
-
Components
-
- Pre-coated, ready to use 96-well strip plate, flat buttom
- Plate sealer for 96 wells
- Reference Standard
- Standard Diluent
- Detection Reagent A
- Detection Reagent B
- Assay Diluent A
- Assay Diluent B
- Reagent Diluent (if Detection Reagent is lyophilized)
- TMB Substrate
- Stop Solution
- Wash Buffer (30 x concentrate)
- Instruction manual
-
-
-
-
Application Notes
-
- Limited by the current condition and scientific technology, we cannot completely conduct the comprehensive identification and analysis on the raw material provided by suppliers. So there might be some qualitative and technical risks to use the kit.
- The final experimental results will be closely related to validity of the products, operation skills of the end users and the experimental environments. Please make sure that sufficient samples are available.
- Kits from different batches may be a little different in detection range, sensitivity and color developing time.
- Do not mix or substitute reagents from one kit lot to another. Use only the reagents supplied by manufacturer.
- Protect all reagents from strong light during storage and incubation. All the bottle caps of reagents should be covered tightly to prevent the evaporation and contamination of microorganism.
- There may be some foggy substance in the wells when the plate is opened at the first time. It will not have any effect on the final assay results. Do not remove microtiter plate from the storage bag until needed.
- Wrong operations during the reagents preparation and loading, as well as incorrect parameter setting for the plate reader may lead to incorrect results. A microplate plate reader with a bandwidth of 10nm or less and an optical density range of 0-3 O.D. or greater at 450 ± 10nm wavelength is acceptable for use in absorbance measurement. Please read the instruction carefully and adjust the instrument prior to the experiment.
- Even the same operator might get different results in two separate experiments. In order to get better reproducible results, the operation of every step in the assay should be controlled. Furthermore, a preliminary experiment before assay for each batch is recommended.
- Each kit has been strictly passed Q.C test. However, results from end users might be inconsistent with our in-house data due to some unexpected transportation conditions or different lab equipments. Intra-assay variance among kits from different batches might arise from above factors, too.
- Kits from different manufacturers for the same item might produce different results, since we have not compared our products with other manufacturers.
-
Comment
-
Information on standard material:
The standard might be recombinant protein or natural protein, that will depend on the specific kit. Moreover, the expression system is E.coli or yeast or mammal cell. There is 0.05% proclin 300 in the standard as preservative.
Information on reagents:
The stop solution used in the kit is sulfuric acid with concentration of 1 mol/L. And the wash solution is TBS. The standard diluent contains 0.02 % sodium azide, assay diluent A and assay diluent B contain 0.01% sodium azide. Some kits can contain is BSA in them.
Information on antibodies:
The provided antibodies and their host vary in different kits. -
Sample Volume
- 100 μL
-
Assay Time
- 3 h
-
Plate
- Pre-coated
-
Protocol
- The test principle applied in this kit is Sandwich enzyme immunoassay. The microtiter plate provided in this kit has been pre-coated with an antibody specific to Interleukin 8 (IL8). Standards or samples are then added to the appropriate microtiter plate wells with a biotin-conjugated antibody specific to Interleukin 8 (IL8). Next, Avidin conjugated to Horseradish Peroxidase (HRP) is added to each microplate well and incubated. After TMB substrate solution is added, only those wells that contain Interleukin 8 (IL8), biotin-conjugated antibody and enzyme-conjugated Avidin will exhibit a change in color. The enzyme-substrate reaction is terminated by the addition of sulphuric acid solution and the color change is measured spectrophotometrically at a wavelength of 450nm ± 10nm. The concentration of Interleukin 8 (IL8) in the samples is then determined by comparing the O.D. of the samples to the standard curve.
-
Reagent Preparation
-
- Bring all kit components and samples to room temperature (18-25 °C) before use. If the kit will not be used up in one time, please only take out strips and reagents for present experiment, and leave the remaining strips and reagents in required condition.
- Standard - Reconstitute the Standard with 0.5 mL of Standard Diluent, keep for 10 minutes at room temperature, shake gently (not to foam). The concentration of the standard in the stock solution is 1,000pg/mL. Please prepare 7 tubes containing 0.25 mL Standard Diluent and produce a double dilution series. Mix each tube thoroughly before the next transfer. Set up 7 points of diluted standard such as 1,000pg/mL, 500pg/mL, 250pg/mL, 125pg/mL, 62.5pg/mL, 31.2pg/mL, 15.6pg/mL, and the last microcentrifuge tube with Standard Diluent is the blank as 0pg/mL.
- Detection Reagent A and Detection Reagent B - If lyophilized reconstitute the Detection Reagent A with 150μL of Reagent Diluent, keep for 10 minutes at room temperature, shake gently (not to foam). Briefly spin or centrifuge the stock Detection A and Detection B before use. Dilute them to the working concentration 100-fold with Assay Diluent A and B, respectively.
- Wash Solution - Dilute 20 mL of Wash Solution concentrate (30x) with 580 mL of deionized or distilled water to prepare 600 mL of Wash Solution (1x).
- TMB substrate - Aspirate the needed dosage of the solution with sterilized tips and do not dump the residual solution into the vial again.
Note:
- Making serial dilution in the wells directly is not permitted.
- Prepare standards within 15 minutes before assay. Please do not dissolve the reagents at 37 °C directly.
- Please carefully reconstitute Standards or working Detection Reagent A and B according to the instruction, and avoid foaming and mix gently until the crystals are completely dissolved. To minimize imprecision caused by pipetting, use small volumes and ensure that pipettors are calibrated. It is recommended to suck more than 10μL for one pipetting.
- The reconstituted Standards, Detection Reagent A and Detection Reagent B can be used only once.
- If crystals have formed in the Wash Solution concentrate (30x), warm to room temperature and mix gently until the crystals are completely dissolved.
- Contaminated water or container for reagent preparation will influence the detection result.
-
Assay Precision
-
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Interleukin 8 (IL8) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Interleukin 8 (IL8) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
-
Restrictions
- For Research Use only
-
-
- by
- Shakti Bioresearch
- No.
- #029582
- Date
- 01/24/2014
- Antigen
- Lot Number
- L131225502
- Method validated
- ELISA
- Positive Control
- Human serum
- Negative Control
- Mouse serum
- Notes
- The kit detected signal in positive control serum and did not detect signal in negative control serum.
- Primary Antibody
- Antigen: Interleukin-8 (IL-8)
- Catalog number: ABIN414887
- Lot number: L131225502
- Secondary Antibody
- Full Protocol
- All reagents in the ELISA kit were brought up to room temperature (RT) before use.
- 100 µl of each sample was added per well to the micro ELISA plate well. All samples and standards were assayed in triplicate.
- The plate was covered with sealer (provided in kit) and incubated for 120 mins at 37°C.
- Liquid was removed from each well by pipette.
- Detection Reagent A was diluted 100 fold in Assay Diluent A. 100 µl of diluted detection reagent A was added to each well and the plate was sealed. The plate was tapped to ensure mixing and incubated for 60 mins at 37°C.
- Wells were washed with 300 µl wash buffer three times. Each wash involved fully aspirating the liquid from each well by pipette. After the last wash the plate was inverted against clean absorbent paper to remove any remaining liquid.
- Detection Reagent B was diluted 100 fold in Assay Diluent B. 100 µl of diluted detection reagent B was added to each well and the plate was sealed. The plate was tapped to ensure mixing and incubated for 30 mins at 37°C.
- Wells were washed with 300 µl wash buffer five times. Each wash involved fully aspirating the liquid from each well by pipette. After the last wash the plate was inverted against clean absorbent paper to remove any remaining liquid.
- 90 µl of Substrate Solution was added to each well and the plate was covered with a new plate sealer. The plate was tapped to ensure mixing and incubated at room temperature in the dark.
- After about 10 mins, when an apparent gradient appeared in the standard wells, the reaction was terminated by adding 50 µl of Stop Solution to each well.
- The optical density (OD value) of each well was read using a micro-plate reader set to 450 nm.
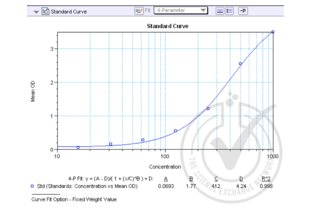
- The triplicate readings for each sample were averaged and the average zero standard optical density subtracted. A standard curve was generated by plotting the mean OD value for each standard on the y-axis against the concentration on the x-axis using Softmax Pro softare.
- The equation y = (A-D)/(1 + (x/C)^B) + D was used to calculate IL-8 concentrations of the samples based on their average OD values.
- Experimental Notes
- Standards at lower level showed higher variability.
- Sensitivity of the assay needs to be improved as the human serum needs to be diluted >10 fold to avoid matrix interference.
Validation #029582 (ELISA)![Successfully validated 'Independent Validation' Badge]()
![Successfully validated 'Independent Validation' Badge]() Validation Images
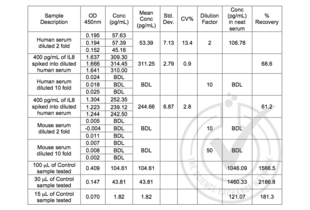
Validation Images![Table 1: ELISA. IL-8 could be detected in human serum (positive control) after 2 fold dilution. Spike controls indicate that there is interference from the human serum matrix and a dilution of >10 fold is required. Mouse serum was used as negative control, there were no detectable levels of IL-8. BDL: below detection limit. Control sample concentration is expected to be 66.78 pg/mL.]() Table 1: ELISA. IL-8 could be detected in human serum (positive control) after 2 fold dilution. Spike controls indicate that there is interference from the human serum matrix and a dilution of >10 fold is required. Mouse serum was used as negative control, there were no detectable levels of IL-8. BDL: below detection limit. Control sample concentration is expected to be 66.78 pg/mL.
Table 1: ELISA. IL-8 could be detected in human serum (positive control) after 2 fold dilution. Spike controls indicate that there is interference from the human serum matrix and a dilution of >10 fold is required. Mouse serum was used as negative control, there were no detectable levels of IL-8. BDL: below detection limit. Control sample concentration is expected to be 66.78 pg/mL.
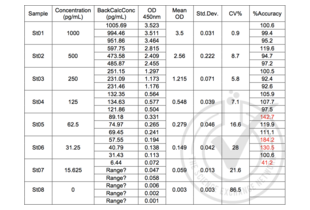
![Table 2: Generation of the Standard Curve. Value for Average Reading is derived from the average of three readings (OD 450nm). The Average Reading for BLANK (0 pg/ml) was subtracted from all Average Readings to yield Average Absorbance values for Standards. Standard deviation is included for all samples. An equation y = (A-D)/(1 + (x/C)^B) + D was generated from the standard curve and used to calculate IL-8 concentrations shown in the Figure 1 and Table 1.]() Table 2: Generation of the Standard Curve. Value for Average Reading is derived from the average of three readings (OD 450nm). The Average Reading for BLANK (0 pg/ml) was subtracted from all Average Readings to yield Average Absorbance values for Standards. Standard deviation is included for all samples. An equation y = (A-D)/(1 + (x/C)^B) + D was generated from the standard curve and used to calculate IL-8 concentrations shown in the Figure 1 and Table 1.
Full Methods
Table 2: Generation of the Standard Curve. Value for Average Reading is derived from the average of three readings (OD 450nm). The Average Reading for BLANK (0 pg/ml) was subtracted from all Average Readings to yield Average Absorbance values for Standards. Standard deviation is included for all samples. An equation y = (A-D)/(1 + (x/C)^B) + D was generated from the standard curve and used to calculate IL-8 concentrations shown in the Figure 1 and Table 1.
Full Methods -
-
Precaution of Use
- The Stop Solution suggested for use with this kit is an acid solution. Wear eye, hand, face, and clothing protection when using this material.
-
Handling Advice
-
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5 % within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end. -
Storage
- 4 °C
-
Storage Comment
-
- For unopened kit: All the reagents should be kept according to the labels on vials. The Standard, Detection Reagent A, Detection Reagent B and the 96-well strip plate should be stored at -20°C upon receipt while the others should be at 4°C.
- For opened kit: When the kit is opened, the remaining reagents still need to be stored according to the above storage condition. Besides, please return the unused wells to the foil pouch containing the desiccant pack, and reseal along entire edge of zip-seal.
Note: It is highly recommended to use the remaining reagents within 1 month provided this is within the expiration date of the kit. - For ELISA kit, 1 day storage at 37°C can be considered as 2 months at 4°C, which means 3 days at 37°C equaling 6 months at 4°C.
-
Expiry Date
- 6 months
-
-
-
: "Study on Adenovirus Infection in vitro with Nanoself-Assembling Peptide as Scaffolds for 3D Culture." in: International journal of nanomedicine, Vol. 15, pp. 6327-6338, (2020) (PubMed).
: "Optimization of Storage Temperature for Retention of Undifferentiated Cell Character of Cultured Human Epidermal Cell Sheets." in: Scientific reports, Vol. 7, Issue 1, pp. 8206, (2019) (PubMed).
: "Polar extract of Curcuma longa protects cartilage homeostasis: possible mechanism of action." in: Inflammopharmacology, Vol. 26, Issue 5, pp. 1233-1243, (2019) (PubMed).
: "Pathogen-dependent modulation of milk neutrophils competence, plasma inflammatory cytokines and milk quality during intramammary infection of Sahiwal (Bos indicus) cows." in: Microbial pathogenesis, Vol. 121, pp. 131-138, (2019) (PubMed).
: "Inhibitory Effect of Low-Intensity Pulsed Ultrasound on the Expression of Lipopolysaccharide-Induced Inflammatory Factors in U937 Cells." in: Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine, Vol. 36, Issue 12, pp. 2419-2429, (2018) (PubMed).
: "CagA promotes proliferation and inhibits apoptosis of GES-1 cells by upregulating TRAF1/4-1BB." in: Molecular medicine reports, Vol. 16, Issue 2, pp. 1262-1268, (2018) (PubMed).
: "The effect of restorative materials on cytokines in gingival crevicular fluid." in: Archives of oral biology, Vol. 84, pp. 139-144, (2018) (PubMed).
: "Inflammatory biomarkers and radiologic measurements in never-smokers with COPD: A cross-sectional study from the CODA cohort." in: Chronic respiratory disease, Vol. 15, Issue 2, pp. 138-145, (2018) (PubMed).
: "Dexmedetomidine attenuates renal fibrosis via α2-adrenergic receptor-dependent inhibition of cellular senescence after renal ischemia/reperfusion." in: Life sciences, Vol. 207, pp. 1-8, (2018) (PubMed).
: "Rho Kinase Type 1 (ROCK1) Promotes Lipopolysaccharide-induced Inflammation in Corneal Epithelial Cells by Activating Toll-Like Receptor 4 (TLR4)-Mediated Signaling." in: Medical science monitor : international medical journal of experimental and clinical research, Vol. 24, pp. 3514-3523, (2018) (PubMed).
: "Mutant DD genotype of NFKB1 gene is associated with the susceptibility and severity of coronary artery disease." in: Journal of molecular and cellular cardiology, Vol. 103, pp. 56-64, (2017) (PubMed).
: "Increased Chondrogenic Potential of Mesenchymal Cells From Adipose Tissue Versus Bone Marrow-Derived Cells in Osteoarthritic In Vitro Models." in: Journal of cellular physiology, Vol. 232, Issue 6, pp. 1478-1488, (2017) (PubMed).
: "Association Between Endothelial Progenitor Cells and Treatment Response in Non-Squamous Non-small Cell Lung Cancer Treated with Bevacizumab." in: Anticancer research, Vol. 37, Issue 10, pp. 5565-5571, (2017) (PubMed).
: "Neutrophil gene dynamics and plasma cytokine levels in dairy cattle during peri-implantation period." in: Veterinary immunology and immunopathology, Vol. 173, pp. 44-9, (2016) (PubMed).
: "A multi-target approach for pain treatment: dual inhibition of fatty acid amide hydrolase and TRPV1 in a rat model of osteoarthritis." in: Pain, Vol. 156, Issue 5, pp. 890-903, (2015) (PubMed).
: "Experimental peri-implant mucositis at different implant surfaces." in: Journal of clinical periodontology, Vol. 41, Issue 5, pp. 513-20, (2014) (PubMed).
: "Expression of interleukine-8 as an independent prognostic factor for sporadic colon cancer dissemination." in: Journal of medicine and life, Vol. 7, Issue 2, pp. 215-9, (2014) (PubMed).
: "Serial cytokine levels during wound healing in rabbit maxillary sinus mucosa." in: Acta oto-laryngologica, Vol. 130, Issue 5, pp. 607-13, (2010) (PubMed).
-
-
- IL-8 (IL8) (Interleukin 8 (IL8))
-
Alternative Name
- IL8
-
Pathways
- TLR Signaling, Cellular Response to Molecule of Bacterial Origin, Regulation of G-Protein Coupled Receptor Protein Signaling, ER-Nucleus Signaling, Hepatitis C, Autophagy
Target See all IL-8 (IL8) ELISA Kits
-

 (18 references)
(18 references) (1 validation)
(1 validation)



