An Introduction to Fluorescence (Part 2)
Written/ Edited by Dr. Ryan Robinson, PhDModern synthetic chemistry, and extensive study of fluorescent materials have given rise to a plethora of new, widely use fluorescent dyes and probes. Today, there are well over 100 different options for researchers that need to label or track a target using fluorescence. These options range from traditional fluorescent dyes like FITC or phycoerithrin, to engineered fluorescent proteins like eGFP or EosFP, to advanced synthetic fluorescent crystals known as quantum dots.
New dyes and probes are engineered for a variety of reasons. Some newer dyes have properties that make them ostensibly better than older options (e.g. they are more photostable, brighter, or have a more narrow emission spectra). Other dyes have been intentionally developed to function best in a specific applications or with a particular piece of instrumentation. However, even amidst a sea of modern options, traditional dyes like FITC and PE often retain a strong following and use. This is due in no small part to researcher familiarity, and the broad availability of inexpensive, proven equipment compatible with these dyes.
Common properties of fluorophores
Fluorescence is among the most extensively studied of all natural phenomena. For most fluorescent probes there are a set of precise measurements that can help a researcher accurately compare the performance of a specific fluorophore. Some of the more commonly referenced criteria are listed below:
- Excitation maxima (ExMax): The wavelength of light most efficiently absorbed by the fluorophore.
- Emission maxima (EmMax): The wavelength of light that will be emitted from the fluorophore at the highest intensity.
Excitation and emission maxima are specific, measured or calculated values describing the wavelength at which a fluorophore most efficiently absorbs or emits light. In reality, most fluorophores both absorb, and emit light in a fairly wide range. The color of light that excites or is emitted from a fluorophore can be interpreted from the excitation and emission maxima. It is also important to note that excitation and emission profiles for a given fluorophore may shift slightly depending on the chemical properties of the environment in which the fluorophore is present (e.g. pH or electrolyte content).
A typical fluorophore excitation and emission profile.
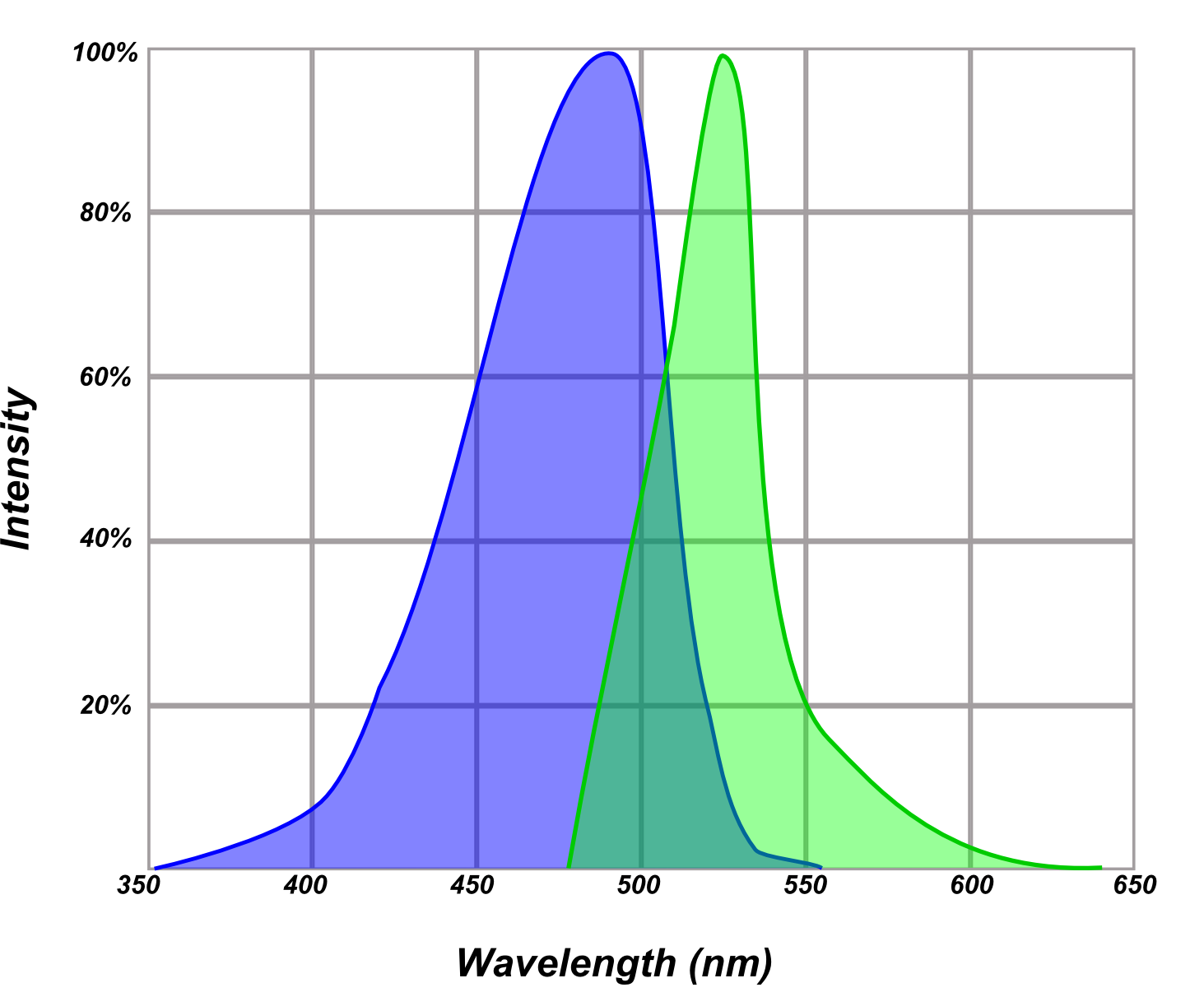
- Extinction coefficient/molar absorption: A measurement of the amount of light of a specific wavelength that can be absorbed by a given material in solution.
- Quantum yield(QY): A measurement of fluorescent efficiency, expressed as a direct ratio of photons emitted to photons absorbed. (i.e. a quantum yield of 0.1 indicates 1 photon is emitted for every 10 photons absorbed).
- Photostability: The ability of a fluorophore to resist photobleaching. Photobleaching is a phenomenon in which a fluorophore will "burn out" and cease to fluoresce after continual, high-intensity excitation.
Extinction coefficient, quantum yield, and photostability all play a role in determining how bright a fluorophore will be. Fluorophores function by absorbing light at one wavelength, and emitting it at another. Fluorophores with a higher quantum yield will emit more light for each photon absorbed. Fluorophores with a higher coefficient of extinction will absorb more light, and will thus emit more light. More photostable fluorophores will remain fluorescent for a longer period of time after continued excitation by an intense source.
ATTO dyes can be used to generate bright, photostable fluorescent signal in a wide variety of sample types and via many different methods. Among the most popular fluorescent dyes on the market today are the ATTO dyes. The image above demonstrates their performance and versatility in a variety of sample types. While they have not yet reached the popularity of the industry standard Alexa Fluor dyes, ATTO dye" conjugates display exceptional versatility, photostability, and brightness, making them the ideal choice for IF and FACS experiments.
Choosing a fluorescent probe
It is worth noting that there is not any single, industry standard for collection of data for fluorescent probes in the life sciences. The criteria listed above may be precisely measured and provided by a manufacturer, or it may not be known. It is also important, when choosing a fluorophore, to try not to get bogged down in the details. The criteria above is not always "all-telling". Perceived brightness by an observer can, at times, differ from calculated brightness values derived from quantum yield and extinction coefficient.
When choosing a fluorescent probe for use in an upcoming experiment it is far more important that the probe chosen is compatible with the design of the experiment, and with the equipment to be used than that it have any specific value associated with the categories listed above.
When choosing a fluorophore, ask yourself these three questions:
"Is this probe compatible with the experiment I've designed?"
Some advanced techniques (e.g. super-resolution-microscopy) are better suited to fluorescent probes with specific characteristics like a bright profile and high photostability. It is always easier to find a product that has a proven track record than to try to press a sub-optimal dye or protein into a service it is unfit for.
"Is this probe compatible with the equipment that I have at my disposal?"
Does the excitation profile of the dye you're choosing match the illumination sources (lasers, Ex filters) that you have available? Does the emission profile of the dye you're choosing match the detection methods that you have available?
"Is this probe compatible with other fluorescent materials in my sample?"
Does the excitation and emission profile of the dye you've chosen overlap with any of the other fluorophores you intend to use in a double labeling experiment? Will you be able to discriminate between signal from your fluorescent probe and background autofluorescence?
Of all of the qualifications that are necessary to consider, it is most important to choose a fluorophore with an excitation and emission profile that is compatible with both the experiment that you intend to use, and the excitation and emission spectra of other fluorescent materials used in your experiment. Double or triple labeling experiments will require particular care to ensure that compatible fluorophores without significant spectral overlap are chosen.
A Note on Autofluorescence:
Many naturally occurring components of the cell will fluoresce at the same wavelength as certain fluorescent dyes and probes. Generally, the problem is trivial as signal from a fluorescent probe will be significantly stronger than background produced by autofluorescence. However, in some instances where the presence of autofluorescent molecules is high, or the signal generated by a fluorescent probe is low this phenomenon can result in difficulty.
To help mitigate confusion from autofluorescence, you may want to consider the following chart of common autofluorescent molecules in the cell:
| Molecule | Emmax(nm) |
|---|---|
| DNA | ~380 |
| NADPH | ~450 |
| Folic Acid | ~450 |
| Retinol | ~515 |
| Flavin | ~540 |
| Chlorophyll | ~673 & ~726 |
If you're still a bit confused about fluorescence, it might be helpful to review the basic details in the prologue to this article. Alternatively, you can always ask one of our technical support scientists. If you'd like information about advanced techniques using fluorescence, like super-resolution microscopy or FRET, make sure to check out advanced fluorescence techniques and tools....coming soon!
Featured RFP Antibodies
- (363)
- (11)
- (1)
- (16)
- (9)
ATTO is a trademark of ATTO-TEC GmbH. Alexa Fluor is a trademark of Life Technologies, Inc.
Additional Reading

Offered pre- and post-sales consultation and support to antibodies-online customers. Provided troubleshooting and technical assistance with proteomics and genomics experiments. Composed and distributed cross-channel digital marketing campaigns.
Go to author page



