SOX2 antibody
Quick Overview for SOX2 antibody (ABIN2855074)
Target
See all SOX2 AntibodiesReactivity
Host
Clonality
Conjugate
Application
Grade
-
-
Cross-Reactivity
- Ceratotherium simum, Frog, Human, Mouse, Rat
-
Characteristics
-
Rabbit Polyclonal antibody to SOX2 (SRY (sex determining region Y)-box 2)
SOX2 antibody [N1C3] -
Purification
- Purified by antigen-affinity chromatography.
-
Immunogen
- Recombinant protein encompassing a sequence within the center region of human SOX2. The exact sequence is proprietary.
-
Isotype
- IgG
-
-
-
-
Application Notes
- WB: 1:5000-1:20000. ICC/IF: 1:100-1:1000. IHC-P: 1:100-1:1000. IHC-Fr: 1:100-1:1000. FACS: 1:50-1:200. IP: 1:100-1:500. CUT&RUN: 1:50. Optimal dilutions/concentrations should be determined by the researcher. Not tested in other applications.
-
Comment
-
Positive Control: Mouse ESC , hESC , NT2D1 , NT2D1
Validation: Comparison, KO/KD, Orthogonal
-
Restrictions
- For Research Use only
-
-
- by
- Mattias Pernebrink and Claudio Cantù; Cantù Lab, Gene Regulation during Development and Disease, Linköping University
- No.
- #104252
- Date
- 05/28/2020
- Antigen
- SOX2
- Lot Number
- 42970
- Method validated
- Cleavage Under Targets and Release Using Nuclease
- Positive Control
- Recombinant anti-H3K27me3 CUT&RUN Positive Control antibody (antibodies-online, ABIN6923144)
- Negative Control
- Monoclonal anti-FLAG (Sigma-Aldrich, F3165)
- Notes
Passed. ABIN2855074 allows for SOX2 targeted digestion using CUT&RUN.
- Primary Antibody
- ABIN2855074
- Secondary Antibody
- Recombinant anti-H3K27me3 CUT&RUN Positive Control antibody (antibodies-online, ABIN6923144)
- Full Protocol
- Cell harvest
- Harvest 500,000 murine neural stem cells from a small thalamic nucleus per antibody to be used at RT.
- Centrifuge cell solution 3 min at 600 x g at RT.
- Remove the liquid carefully.
- Gently resuspend cells in 1 mL Wash Buffer (20 mM HEPES pH 7.5, 150 mM NaCl, 0.5 mM Spermidine, Roche Complete Protease Inhibitor EDTA-free) by pipetting and transfer cell solution to a 2 mL microcentrifuge tube.
- Centrifuge cell solution 3 min at 600 x g at RT and discard the supernatant.
- Repeat twice for a total of three washes.
- Resuspend cell pellet in 1 mL Wash Buffer by gently pipetting.
- Concanavalin A beads preparation
- Prepare one 1.5 mL microcentrifuge tube.
- Gently resuspend the magnetic Concanavalin A Beads (antibodies-online, ABIN6923139).
- Pipette 10 µL Con A Beads slurry for each sample into the 1.5 mL microcentrifuge tube.
- Place the tube on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tube from the magnetic stand.
- Pipette 1 mL Binding Buffer (20 mM HEPES pH 7.5, 10 mM KCl, 1 mM CaCl2, 1 mM MnCl2) into each tube and resuspend ConA beads by gentle pipetting.
- Spin down the liquid from the lid with a quick pulse in a table-top centrifuge.
- Place the tubes on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tube from the magnetic stand.
- Repeat twice for a total of three washes.
- Gently resuspend the ConA Beads in a volume of Binding Buffer corresponding to the original volume of bead slurry, i.e. 10 µL per sample.
- Cell immobilization – binding to Concanavalin A beads
- Carefully vortex the cell suspension and add 10 µL of the Con A beads in Binding Buffer to the cell suspension for each sample.
- Close tube tightly and rotate for 10 min at RT.
- Cell permeabilization and primary antibody binding
- Divide cell suspension into separate 2 mL microcentrifuge tubes, one for each antibody (500,000 cells per sample).
- Place the microcentrifuge tubes on a magnetic stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tubes from the magnetic stand.
- Place each tube at a low angle on the vortex mixer set to a low speed and add 150 µL Digitonin Wash buffer (wash buffer with 0.025% (wt/vol) Digitonin) supplemented with 2 mM EDTA.
- Gently vortex the microcentrifuge tubes until the beads are resuspended.
- Add 1.5 µL antibody (anti-SOX2, positive control, and negative control) to the respective tube, corresponding to a 1:100 dilution.
- Rotate the microcentrifuge tubes ON at 4 °C.
- Spin down the liquid and place the tubes on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tubes from the magnetic stand.
- Resuspend with 1 mL Digitonin Wash Buffer and mix by inversion. If clumping occurs, gently remove the clumps with a 1 ml pipette tip.
- Repeat once for a total of two washes.
- pA-MNase Binding
- Place the tubes on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tubes from the magnetic stand.
- Vortex the sample at low speed and add 150 μL pA-MNase solution at 700 ng/mL (1:200 dilution of a 140 µg/mL glycerol stock in Digitonin Wash Buffer) per sample, gently resuspending the beads by pipetting.
- Rotate the microcentrifuge tubes for 1 h at 4 °C.
- Spin down the liquid and place the tubes on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Remove the microcentrifuge tubes from the magnetic stand.
- Resuspend with 1 mL Digitonin Wash Buffer and mix by inversion. If clumping occurs, gently remove the clumps with a 1 mL pipette tip.
- Repeat once for a total of two washes.
- MNase digestion and release of pA-MNase-antibody-chromatin complexes
- Spin down the liquid from the lid with a quick pulse in a table-top centrifuge.
- Place the tubes on a magnet stand until the fluid is clear. Remove the liquid carefully.
- Place each tube at a low angle on the vortex mixer set to a low speed and add 100 μL Digitonin Wash buffer per sample along the side of the tube.
- Place tubes in a heat block, kept on ice, and allow to chill.
- Add 2 μL 0.1 M CaCl2 to each sample.
- Incubate tubes at 0 °C for 30 min.
- Add 100 μL 2xSTOP buffer (340 mM NaCl, 20 mM EDTA, 4 mM EGTA, 0.05% (wt/vol) Digitonin, 100 μg/mL RNAse A, 50 μg/mL Glycogen).
- Incubate tubes at 37 °C for 30 min.
- Place the tubes on a magnet stand until the fluid is clear.
- Transfer the supernatant containing the pA-MNase-bound digested chromatin fragments to fresh 1.5 mL microcentrifuge tubes.
- DNA extraction
- Add 2 µL 10% SDS to a final concentration of 0.1% and 2.5 µL Proteinase K (20 mg/mL) to each supernatant.
- Gently vortex tubes at a low speed of approximately 1,100 rpm.
- Incubate tubes at 50 °C for 1 h.
- Add 200 µL PCI to tube.
- Vortex tubes thoroughly at high speed until the liquid appears milky.
- Centrifuge tubes in a tabletop centrifuge at 16,000 x g at RT for 5 min.
- Carefully transfer the upper aqueous phase to a fresh 1.5 mL microcentrifuge tube containing 200 µL chloroform:isoamyl alcohol 24:1.
- Vortex tubes thoroughly at high speed until the liquid appears milky.
- Centrifuge tubes in a tabletop centrifuge at 16,000 x g at 4 °C for 5 min.
- Carefully transfer to upper aqueous phase to a fresh 1.5 mL microcentrifuge tube containing 2 µL glycogen (diluted 1:10 to 2 mg/mL from the 20 mg/mL stock solution).
- Add 20 µL 3 M NaOAc pH 5.2.
- Add 400 µL 100% ethanol.
- Place tubes for at -20 °C ON.
- Centrifuge tubes in a tabletop centrifuge at 16,000 x g at 4 °C for 5min.
- Remove the liquid carefully with a pipette.
- Wash pellet with 1ml 70% ethanol.
- Centrifuge tubes in a tabletop centrifuge at 16,000 x g at 4 °C for 1 min.
- Remove the liquid carefully with a pipette.
- Air-dry the pellet, then dissolve in 30 µL 1 mM Tris-HCl, 0.1 mM EDTA.
- Library preparation and sequencing
- Libraries were prepared using KAPA HyperPrep Kit using KAPA Dual-Indexed adapters according to protocol.
- Samples were sequenced on an Illumina NextSeq 500 sequencer, using a NextSeq 500/550 High Output Kit v2.5 (75 Cycles), 36bp PE.
- Read mapping and Peak calling
- Reads were mapped to the GRCm38 (mm10) mouse genome using Bowtie2 with options: --local --very-sensitive- local --no-unal --no-mixed --no-discordant.
- Peaks were called using MACS2 with options -f BAMPE --keep-dup all –nomodel.
- Experimental Notes
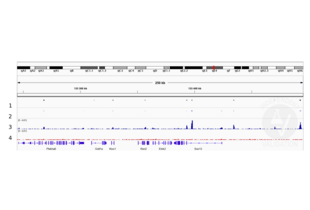
Peaks generated using ABIN2855074 for CUT&RUN aligned well with Sox2 ChIP-seq tracks in neural stem cells (Bertolini et al., 2019).
The experiments were done on mouse tissue, using a small thalamic nucleus inside the brain. These are very valuable tissues composed by small numbers of cells that also express very low levels of this TF. Accordingly, the assay is particularly challenging and the success more indicative of a good antibody.
Sequencing depth was 0.25x coverage for the SOX2 sample and 0.15x coverage for H3K27me3, with coverage being defined as number of bases sequenced/genome size. Assuming a genome size of 2.5x109 this corresponds to 625x106 and 375x106 bases sequenced, i.e. 8.68x106 and 5.21x106 36 bp PE reads respectively.
Validation #104252 (Cleavage Under Targets and Release Using Nuclease)![Successfully validated 'Independent Validation' Badge]()
![Successfully validated 'Independent Validation' Badge]() Validation Images
Validation Images![Alignment tracks from CUT&RUN targeting SOX2 in neural stem cells. 1 Peak track from ChIP-seq targeting Sox2 in neural stem cells (Bertolini et al., 2019). 2. Peaks called by MACS2 from CUT&RUN data using anti-SOX2 antibody ABIN2855074. 3, Alignment track for CUT&RUN reads obtained using snti-SOX2 antibody ABIN2855074 in neural stem cells. 4. Alignment track for negative control. Reads are normalized to sequencing depth per million reads.]() Alignment tracks from CUT&RUN targeting SOX2 in neural stem cells. 1 Peak track from ChIP-seq targeting Sox2 in neural stem cells (Bertolini et al., 2019). 2. Peaks called by MACS2 from CUT&RUN data using anti-SOX2 antibody ABIN2855074. 3, Alignment track for CUT&RUN reads obtained using snti-SOX2 antibody ABIN2855074 in neural stem cells. 4. Alignment track for negative control. Reads are normalized to sequencing depth per million reads.
Full Methods
Alignment tracks from CUT&RUN targeting SOX2 in neural stem cells. 1 Peak track from ChIP-seq targeting Sox2 in neural stem cells (Bertolini et al., 2019). 2. Peaks called by MACS2 from CUT&RUN data using anti-SOX2 antibody ABIN2855074. 3, Alignment track for CUT&RUN reads obtained using snti-SOX2 antibody ABIN2855074 in neural stem cells. 4. Alignment track for negative control. Reads are normalized to sequencing depth per million reads.
Full Methods -
-
Format
- Liquid
-
Concentration
- 0.15 mg/mL
-
Buffer
- 1XPBS ( pH 7), 1 % BSA, 20 % Glycerol, 0.025 % ProClin 300
-
Preservative
- ProClin
-
Precaution of Use
- This product contains ProClin: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
-
Storage
- 4 °C,-20 °C
-
Storage Comment
- Store as concentrated solution. Centrifuge briefly prior to opening vial. For short-term storage (1-2 weeks), store at 4°C. For long-term storage, aliquot and store at -20°C or below. Avoid multiple freeze-thaw cycles.
-
-
-
: "SOX2 and NR2F1 coordinate the gene expression program of the early postnatal visual thalamus." in: Biology open, Vol. 14, Issue 8, (2025) (PubMed).
: "Sox2 controls neural stem cell self-renewal through a Fos-centered gene regulatory network." in: Stem cells (Dayton, Ohio), (2021) (PubMed).
: "FOS Rescues Neuronal Differentiation of Sox2-Deleted Neural Stem Cells by Genome-Wide Regulation of Common SOX2 and AP1(FOS-JUN) Target Genes." in: Cells, Vol. 10, Issue 7, (2021) (PubMed).
: "Function of BMP4 in the Formation of Vasculogenic Mimicry in Hepatocellular Carcinoma." in: Journal of Cancer, Vol. 11, Issue 9, pp. 2560-2571, (2020) (PubMed).
: "lncRNA 5430416N02Rik Promotes the Proliferation of Mouse Embryonic Stem Cells by Activating Mid1 Expression through 3D Chromatin Architecture." in: Stem cell reports, Vol. 14, Issue 3, pp. 493-505, (2020) (PubMed).
: "Involvement of collagen XVII in pluripotency gene expression and metabolic reprogramming of lung cancer stem cells." in: Journal of biomedical science, Vol. 27, Issue 1, pp. 5, (2020) (PubMed).
: "ERK Activation Modulates Cancer Stemness and Motility of a Novel Mouse Oral Squamous Cell Carcinoma Cell Line." in: Cancers, Vol. 12, Issue 1, (2019) (PubMed).
: "Compromised Chondrocyte Differentiation Capacity in TERC Knockout Mouse Embryonic Stem Cells Derived by Somatic Cell Nuclear Transfer." in: International journal of molecular sciences, Vol. 20, Issue 5, (2019) (PubMed).
: "The Sox2 promoter-driven CD63-GFP transgenic rat model allows tracking of neural stem cell-derived extracellular vesicles." in: Disease models & mechanisms, Vol. 11, Issue 1, (2018) (PubMed).
: "Enforcement of developmental lineage specificity by transcription factor Oct1." in: eLife, Vol. 6, (2018) (PubMed).
: "Activation of Sphingosine 1-Phosphate Receptor 1 Enhances Hippocampus Neurogenesis in a Rat Model of Traumatic Brain Injury: An Involvement of MEK/Erk Signaling Pathway." in: Neural plasticity, Vol. 2016, pp. 8072156, (2017) (PubMed).
: "Suberoylanilide hydroxamic acid represses glioma stem-like cells." in: Journal of biomedical science, Vol. 23, Issue 1, pp. 81, (2017) (PubMed).
-
-
- SOX2 (SRY (Sex Determining Region Y)-Box 2 (SOX2))
-
Alternative Name
- SRY-box 2
-
Background
-
This intronless gene encodes a member of the SRY-related HMG-box (SOX) family of transcription factors involved in the regulation of embryonic development and in the determination of cell fate. The product of this gene is required for stem-cell maintenance in the central nervous system, and also regulates gene expression in the stomach. Mutations in this gene have been associated with optic nerve hypoplasia and with syndromic microphthalmia, a severe form of structural eye malformation. This gene lies within an intron of another gene called SOX2 overlapping transcript (SOX2OT).
Cellular Localization: Nucleus -
Molecular Weight
- 34 kDa
-
Gene ID
- 6657
-
UniProt
- P48431
-
Pathways
- Dopaminergic Neurogenesis, Sensory Perception of Sound, Stem Cell Maintenance, Cell RedoxHomeostasis
Target
-

 (12 references)
(12 references) (1 validation)
(1 validation)



