Caspase 8 Antibodies
Your search for reliable Caspase 8 antibodies ends here. Caspase 8, known by aliases such as casp8.L, LOC661095, CASP8, LOC5563818, casp8, LOC100222284, Casp8, is an integral part of our antibody range. Whether you're working with Human, Mouse, Rat, Monkey, Pig, or other species, our range of Caspase 8 antibodies offer precise detection across diverse samples. These specialized antibodies are tailored for various scientific applications like WB, ELISA, IHC, FACS, IF (cc), providing you with options like polyclonal, recombinant, and monoclonal antibodies, sourced from different host species such as Rabbit, Mouse, Rat. The efficacy of our antibodies is well-established, demonstrated through multiple methods.
Detailed information, including references, images, and validations by other customers, can be found on each product page. Should you require assistance in finding a specific product, our customer service team is ready to assist. Utilize our Caspase 8 antibodies in your research endeavors for dependable Caspase 8 detection.
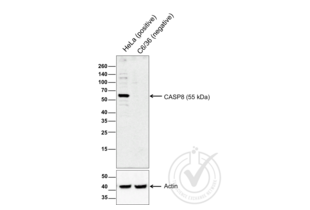
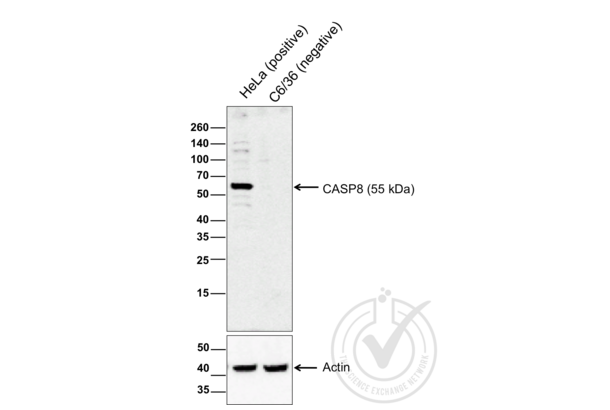 Caspase 8 antibody (AA 411-482) (ABIN724205)
Caspase 8 antibody (AA 411-482) (ABIN724205)
CASP8 Reactivity: Human, Mouse, Rat, Monkey, Pig, Sheep WB, ELISA, IHC (p), FACS, IF (cc), IF (p), IHC (fro) Host: Rabbit Polyclonal unconjugated
CASP8 Reactivity: Human WB, IHC, IF Host: Rabbit Polyclonal unconjugated
CASP8 Reactivity: Human WB, IHC, IP, ICC Host: Rabbit Polyclonal unconjugated
Caspase 8 Antibodies by Grade
Find Caspase 8 Antibodies with a specific Grade. The Grade listed below are among those available. Click on a link to go to the corresponding products.
Caspase 8 Antibodies by Reactivity
Find Caspase 8 Antibodies for a variety of species such as anti-Human Caspase 8, anti-Mouse Caspase 8, anti-Rat Caspase 8. The species listed below are among those available. Click on a link to go to the corresponding products.
Caspase 8 Antibodies by Host
Find Caspase 8 Antibodies with a specific Host. The Host listed below are among those available. Click on a link to go to the corresponding products.
Caspase 8 Antibodies by Clonality
Find available monoclonal or polyclonal Caspase 8 Antibodies. Click on a link to go to the corresponding products.
Popular Caspase 8 Antibodies
- (15)
- (5)
- (1)
- (17)
- (12)
- (10)
- (9)
- (9)
- (4)
- (5)
- (2)
- (5)
- (9)
- (6)
- (4)
- (4)
- (4)
- (4)
- (3)
- (3)
- (2)
- (2)
Latest Publications for our Caspase 8 Antibodies
: "NLRP12 collaborates with NLRP3 and NLRC4 to promote pyroptosis inducing ganglion cell death of acute glaucoma." in: Molecular neurodegeneration, Vol. 15, Issue 1, pp. 26, (2020) (PubMed).: "The protective role of the MKP-5-JNK/P38 pathway in glucolipotoxicity-induced islet β-cell dysfunction and apoptosis." in: Experimental cell research, Vol. 382, Issue 1, pp. 111467, (2020) (PubMed).
: "Downregulation of METTL14 increases apoptosis and autophagy induced by cisplatin in pancreatic cancer cells." in: The international journal of biochemistry & cell biology, Vol. 122, pp. 105731, (2020) (PubMed).
: "Triggering apoptosis by oroxylin A through caspase-8 activation and p62/SQSTM1 proteolysis." in: Redox biology, Vol. 29, pp. 101392, (2020) (PubMed).
: "Emodin induced necroptosis in the glioma cell line U251 via the TNF-α/RIP1/RIP3 pathway." in: Investigational new drugs, Vol. 38, Issue 1, pp. 50-59, (2020) (PubMed).
: "Microglia-derived TNF-α mediates endothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke." in: Cell death & disease, Vol. 10, Issue 7, pp. 487, (2020) (PubMed).
: "GL-V9 exerts anti-T cell malignancies effects via promoting lysosome-dependent AKT1 degradation and activating AKT1/FOXO3A/BIM axis." in: Free radical biology & medicine, Vol. 145, pp. 237-249, (2020) (PubMed).
: "Xanthones from the Bark of Garcinia xanthochymus and the Mechanism of Induced Apoptosis in Human Hepatocellular Carcinoma HepG2 Cells via the Mitochondrial Pathway." in: International journal of molecular sciences, Vol. 20, Issue 19, (2020) (PubMed).
: "Histone methyltransferase SETDB1 promotes colorectal cancer proliferation through the STAT1-CCND1/CDK6 axis." in: Carcinogenesis, Vol. 41, Issue 5, pp. 678-688, (2020) (PubMed).
: "Giardia duodenalis induces extrinsic pathway of apoptosis in intestinal epithelial cells through activation of TNFR1 and K63 de-ubiquitination of RIP1 in vitro." in: Microbial pathogenesis, Vol. 149, pp. 104315, (2020) (PubMed).
Aliases for Caspase 8 Antibodies
caspase 8 L homeolog (casp8.L) Antibodiesdeath related ced-3/Nedd2-like protein (LOC661095) Antibodies
caspase 8 (CASP8) Antibodies
caspase-8 (LOC5563818) Antibodies
caspase-8 (casp8) Antibodies
caspase-8-like (LOC100222284) Antibodies
caspase 8 (Casp8) Antibodies
caspase 8, apoptosis-related cysteine peptidase (casp8) Antibodies
ALPS2B Antibodies
CAP4 Antibodies
CASP-8 Antibodies
Casp-8 Antibodies
casp8 Antibodies
CASP8 Antibodies
casp8-A Antibodies
caspase-8 Antibodies
FLICE Antibodies
MACH Antibodies
Mch5 Antibodies
MCH5 Antibodies
xCaspase-8 Antibodies
zgc:92075 Antibodies
Did you look for something else?
- Caspase 7 Antibodies
- Caspase 6 Antibodies
- Caspase 4 Antibodies
- Caspase 3 Antibodies
- Caspase 2 Antibodies
- Caspase 12 (Gene/pseudogene) Antibodies
- Caspase 10 Antibodies
- Caspase 1 Antibodies
- CASP5 Antibodies
- CASP14 Antibodies
- CasLTR2 Antibodies
- CASKIN2 Antibodies
- CASK Antibodies
- Casein Kinase 1, delta Antibodies
- Casein Kinase 1 gamma 2 Antibodies
- Casein alpha-S2 Antibodies
- Casein alpha S1 Antibodies
- Casein Antibodies
- CASC5 Antibodies
- CASC4 Antibodies
- Caspase 9 Antibodies
- Caspase Activity and Apoptosis Inhibitor 1 Antibodies
- CASQ2 Antibodies
- CASR Antibodies
- CASS4 Antibodies
- CASZ1 Antibodies
- Catalase Antibodies
- Catechol-O-Methyltransferase Antibodies
- Cathelicidin Antibodies
- Cathepsin 7 Antibodies
- Cathepsin B Antibodies
- Cathepsin D Antibodies
- Cathepsin E Antibodies
- Cathepsin F Antibodies
- Cathepsin G Antibodies
- Cathepsin H Antibodies
- Cathepsin K Antibodies
- Cathepsin L1 Antibodies
- Cathepsin L2 Antibodies
- Cathepsin S Antibodies




