An Introduction to ELISA (Part 2)
Written/ Edited by Dr. Stefan Pellenz, PhDELISA is an exceptionally powerful and versatile technique, and there are number of different ways to approach it. This article covers the various ELISA formats that are most commonly commercially available, and discusses the nuanced differences between them.
If you're looking for a basic general overview of the ELISA, and would like to know whether an ELISA is the right assay design for your experiment, please see An Introduction to ELISA (Part 1).
Direct ELISA
The direct ELISA is the simplest ELISA format. In a direct ELISA:
- The analyte is immobilized on a solid support (most commonly a 96-well microtiter plate).
- An enzyme conjugated antibody directed against the antigen is washed over the plate.
- The antibody recognizes and binds the immobilized analyte, any unbound antibody is rinsed away.
- A colorless chromogenic substrate like TMB (3,3',5,5'-tetramethlybenzidene) is introduced to the solution.
- The enzyme conjugate reacts with the chromogenic substrate, converting it into a dark colored derivative.
- The degree of color change is measured on a spectrophotometric plate reader.
Looking for high-quality ELISA kits?
We offer reliable ELISA kits for more than 3,000 different targets with references, images, validation data.
Discover our ELISA kit portfolioIt is important to remember that the enzyme catalyzing the color-change reaction is conjugated to an antibody, which is in turn bound to antigen which has been immobilized on the plate. Therefore, the rate that the reaction occurs is dependent upon the amount of enzyme present, which is in turn dependent upon the quantity of analyte bound to the plate and the affinity of the antibody for said analyte.
Note: Left unchecked, any quantity of enzyme would eventually catalyze nearly all of the TMB in solution, negating the ability to differentiate between samples with a high concentration of analyte and those with a low concentration of analyte. To prevent this, the reaction is stopped at a predetermined time-point by addition of sulfuric acid.
Direct ELISAs are seldom commercially available, principally because the primary concern of most end users is confirming the presence and measuring the concentration of their analyte in a complex biological sample. The nature of the matrix in these samples often precludes the researcher from affixing the sample to a plate or other solid support. A researcher might choose to use a direct format ELISA if they wanted to test the efficacy of a new antibody against a known target that they could immobilize on a microtiter plate, or if she wanted to compare the concentration of a purified recombinant protein against a set of known concentration standards.
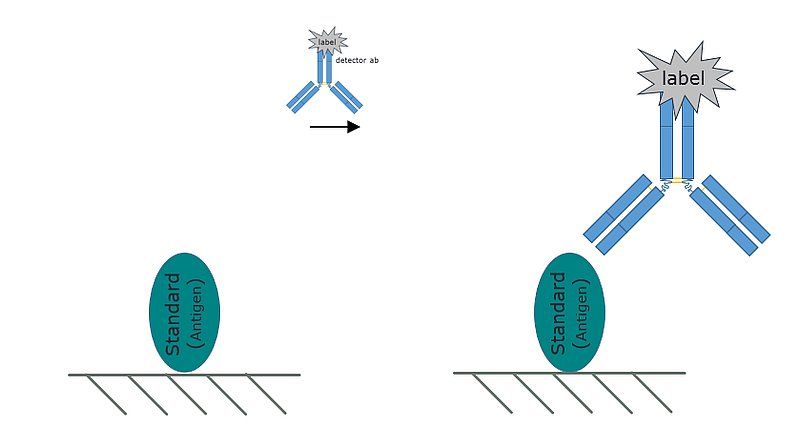
Fig. 1: A representative image of the direct ELISA. The analyte is coated directly on the plate, and is recognized by an enzyme conjugated antibody.
Indirect ELISA
The indirect ELISA is a variant of the direct ELISA. The process is identical, save for the detection step. In an indirect ELISA the primary antibody used to detect the analyte is not conjugated to an enzyme. Instead, an enzyme-conjugated secondary antibody which is reactive against the primary antibody is added to the mixture.
Note: Indirect ELISA is also used to detect and quantify seropositivity for a specific antigen. For example ABIN1540248 uses indirect ELISA method to detect antibodies against the drug Infliximab in human serum. A known quantity of Infliximab is coated on the plate, and added to the coated wells. A labeled secondary antibody is then used to detect the human antibdodies against Infliximab which have been captured by the immobilized antigen.
Use of a secondary antibody has many of the same advantages in an ELISA as in other proteomics methods. In addition to not requiring the researcher to undergo the sometimes arduous process of conjugating a primary antibody, a secondary antibody can help to amplify a weak signal, allowing a previously undetectable signal to be visualized and lowering the detection threshold of the assay in question.
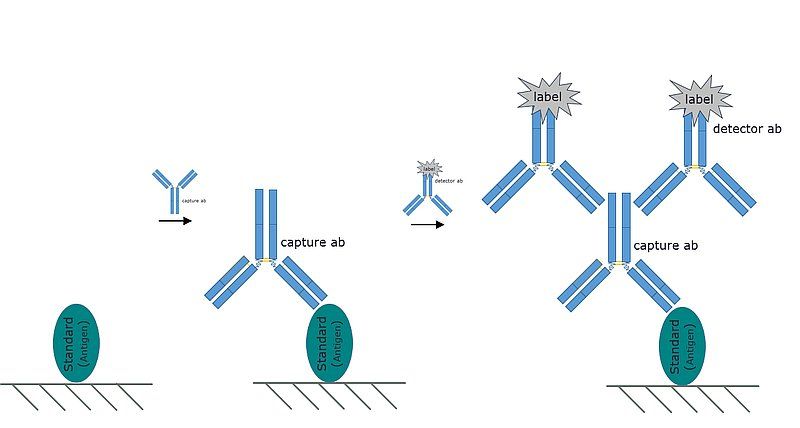
Fig. 2: A representative image of the indirect ELISA. The analyte is coated directly on the plate, however the primary antibody recognizing the antigen is not labeled. Instead a labeled secondary antibody, reactive against the primary, is used.
Sandwich (capture) ELISA
Most commercially available ELISAs are sandwich format assays. The sandwich format assay is so named because the analyte is “sandwiched” two different antibodies. In a sandwich format assay:
- The analyte is NOT directly immobilized on the plate.
- A "capture" antibody is immobilized on the surface of the wells of the plate.
- The "capture" antibody binds and retains analyte from the sample. The remaining matrix is rinsed away.
- An enzyme conjugated "detector" antibody, raised against a different epitope on the analyte is added to the plate.
- The standard colorimetric detection method (described above) is used to detect and quantify analyte in the sample.
Note: antibodies-online offers hundreds of approved antibodies for
and in a sandwich format assay. However, if the option is available you may want to consider purchasing an approved antibody pair to ensure compatibility when developing a sandwich format assay de novo.A monoclonal antibody is often chosen as a capture antibody in a sandwich format ELISA. Monoclonal antibodies, by virtue of their nature, bind only a single epitope on an antigen. This commonly serves to increase specificity of an assay and reduce background noise, making them an attractive choice for assay design.
Employing monoclonal antibodies for both capture and detection in a sandwich format assay is generally not advised. Unless the epitope recognized by both the capture and detection antibodies have been precisely mapped, it is difficult or impossible to predict whether the capture antibody will block binding of the detection antibody due to steric hindrance. The binding constant of any individual monoclonal is also commonly lower than that of the best binders in a heterogeneous mixture of polyclonal antibody, so an assay designed exclusively with monoclonal antibodies will likely never be as sensitive as one employing at least one polyclonal.
As with other formats, detection in a sandwich format assay can be either direct or indirect (i.e. the detection antibody may be directly labeled, or one may employ a conjugated secondary antibody, reactive against the detection antibody). If an indirect detection method is chosen, special care must be taken to ensure that the secondary antibody used reacts only with the deetection antibody and not with the capture antibody coated on the plate.
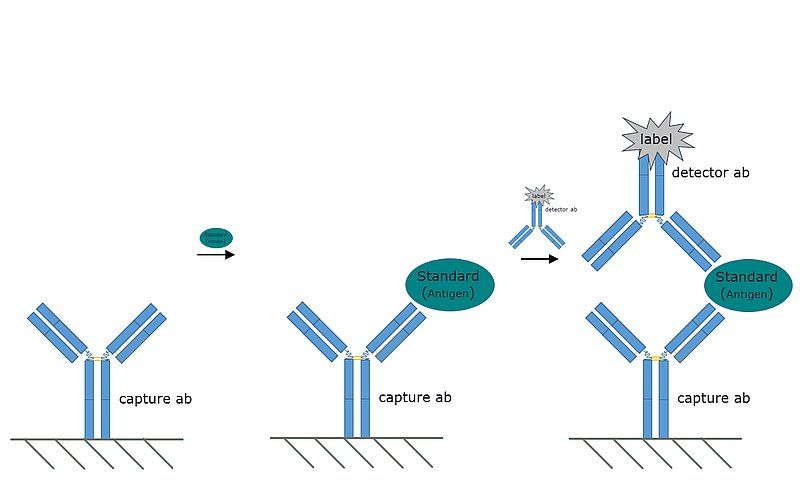
Fig. 3: A representative image of the Sandwich format ELISA. The sandwich format is so named because the analyte is "sandwiched" between a capture and detection antibody.
Competitive ELISA
The competitive format ELISA is, perhaps, the most difficult assay design to comprehend. In a competitive format assay, as antigen concentration in a sample increases, signal intensity decreases. In a competitive ELISA:
- Antigen standard is immobilized on the surface of a plate.
- Antibody against the analyte is incubated with the sample.
- Analyte in the sample binds and occupies antibody in solution.
- The antibody/sample mixture is washed over the antigen coated wells.
- Unoccupied antibodies (those that have not bound analyte in the sample) will bind the antigen standard on the plate.
- Occupied antibodies (those that have bound analyte in the sample) are rinsed away.
- A standard colorimetric detection method is used to detect and quantify analyte in the sample.
The central concept behind a competitive ELISA is that a larger quantity of analyte in a sample results in fewer free antibodies in solution and by extension a smaller number of labeled antibodies bound to the standard on the plate and a less intense signal.
The principal advantage of a competitive ELISA, when compared to a sandwich format assay, is that only one antibody needs to bind the analyte for detection. This makes the competitive assay an attractive option if no suitable antibody pair can be identified for sandwich ELISA, or if the analyte in question is too small to permit binding of both a capture and detection antibody.
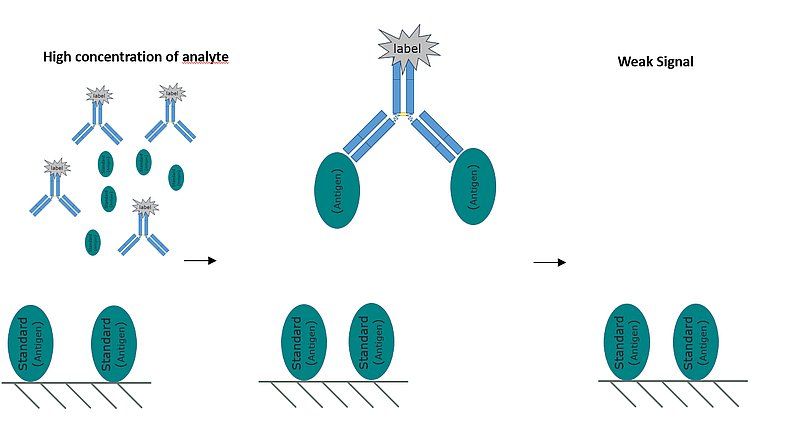
Fig. 4: A representative image of the competitive format ELISA. In a competitive ELISA, a higher concentration of analyte in the sample results in a reduced signal.
Competitive Inhibition ELISA
Most commercially available competitive ELISA kits (including those sold at antibodies-online) follow the competitive inhibition format. Competitive inhibition ELISAs differ slightly from a classical competitive ELISA. In a competitive inhibition assay, a capture antibody is coated on a 96-well plate (much like a sandwich-format ELISA). Unlabeled antigen in the sample, and a known concentration of labeled antigen standard (conjugated to HRP or a similar detection mechanism) then compete for binding affinity of the immobilized antibody.
In a competitive inhibition ELISA, as in a classical competitive ELISA, signal derived from color change is inversely proportional to the amount of antigen in the sample. A large amount of antigen present in the sample results in less labeled standard bound to antibody on the plate, and a lower amount of signal. Conversely, a small amount of antigen in the sample allows for a greater quantity of labeled standard to bind the plate, and produces a higher colorimetric signal.
Conclusion
ELISA is a rapid, simple, and powerful method, offering a host of advantages over other common proteomics techniques. Hopefully, this document has given you some insight the different commercially available ELISA options, and helped you to decide whether ELISA is the right method for your intended experiment. If you still have questions about this or any other method, please feel free to ask one of our technical support scientists. They would be happy to help you find the right product to fit your needs.
antibodies-online.com offers the world's largest selection of ELISA kits and research reagents, distributing hundreds of thousands of high-quality, proven kits from suppliers like Raybiotech, Assay Biotechnology, and Arbor Assays. We also offer a wide variety of products approved for ELISA-like and ELISA-derived methods that extend beyond the scope of this article. The antibodies-online catalog contains a plethora for applications like ELISpot and CLIA (a highly sensitive ELISA-like technique that uses chemiluminescence as a detection method). Contact us to learn more about these methods.
Selection of our most popular ELISA kit products
- (24)
- (3)
- (1)
- (25)
- (6)
- (13)
- (6)
- (1)
- (1)
- (1)

Goal-oriented, time line driven scientist, proficiently trained in different academic institutions in Germany, France and the USA. Experienced in the life sciences e-commerce environment with a focus on product development and customer relation management.
Go to author page



