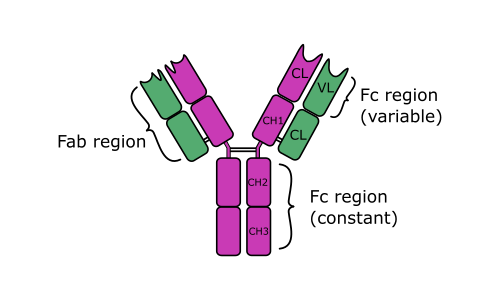
IgD (IgD) is a monomeric antibody isotype that is expressed in the plasma membranes of immature B-lymphocytes. IgD is also produced in a secreted form that is found in small amounts in blood serum. Secreted IgD is made up of two heavy chains of the delta class, and two light chains. IgD's function has long been a conundrum and is still incompletely understood. IgD is present in species ranging from fish to human (probably with the exception of birds), indicating that IgD has been preserved since its ancient origins which suggests that IgD has important immunological functions.
IgD Secondary Antibodies
- (6)
- (2)
- (3)
- (6)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (5)
- (1)
- (1)
Featured IgD Antibodies conjugated to Alexa Fluor 700
Featured IgD Antibodies conjugated to Alexa Fluor 647
Featured IgD Antibodies conjugated to Alexa Fluor 488
Featured IgD Antibodies conjugated to FITC
- (1)
- (1)
- (1)
- (1)
What is IgD?
IgD (IgD) is a monomeric antibody isotype that is expressed in the plasma membranes of immature B-lymphocytes. IgD is also produced in a secreted form that is found in small amounts in blood serum. Secreted IgD is made up of two heavy chains of the delta class, and two light chains.
What is the function of IgD?
IgD's function has long been a conundrum and is still incompletely understood. IgD is present in species ranging from fish to human (probably with the exception of birds), indicating that IgD has been preserved since its ancient origins which suggests that IgD has important immunological functions. It is known that IgD signals for B cells to be activated in order for them to participate in the immune defence. Apparently IgM and IgD share this role, with IgM being expressed sooner in the life-cycle of B-cells than IgD.
Learn more about other secondary antibody types

Creative mind of antibodies-online with a keen eye for details. Proficient in the field of life-science with a passion for plant biotechnology and clinical study design. Responsible for illustrated and written content at antibodies-online as well as supervision of the antibodies-online scholarship program.
Go to author page



