Western Blotting (Immunoblot): Gel Electrophoresis for Proteins
Written/ Edited by Dr. Stefan Pellenz, PhDWestern Blotting (also called immunoblotting) is a technique used for analysis of individual proteins in a protein mixture (e.g. a cell lysate). In Western blotting (immunoblotting) the protein mixture is applied to a gel electrophoresis in a carrier matrix (SDS-PAGE, native PAGE, isoelectric focusing, 2D gel electrophoresis, etc.) to sort the proteins by size, charge, or other differences in individual protein bands. The separated protein bands are then transferred to a carrier membrane (e.g. nitrocellulose, nylon or PVDF). This process is called blotting. The proteins adhere to the membrane in the same pattern as they have been separated due to interactions of charges. The proteins on this immunoblot are then accessible for antibody binding for detection.
Antibodies are used to detect target proteins on the western blot (immunoblot). The antibodies are conjugated with fluorescent or radioactive labels or enzymes that give a subsequent reaction with an applied reagent, leading to a coloring or emission of light, enabling detection.
The term Western Blotting is based on a play of words. The southern blot, which is a method to detect specific DNA sequences, is named after Ed Southern, who first described this procedure. The western blot (immunoblot), as well as the northern blot (for RNA detection), play on the meaning of this name.
Different Types of Gel Electrophoresis for Proteins
One can choose from different types of gel electrophoresis for proteins depending on the criteria by which the proteins should be separated. Some commonly used electrophoretic methods are: SDS-PAGE, native-PAGE and isoelectric focusing.
SDS-PAGE:
This is a denaturing method as it treats the proteins with anionic SDS detergent (sodiumdodcylsulfate). Secondary- and tertiary structure are destroyed by this process. Additionally, SDS binds the proteins and thereby covers their chemical charges, leading to equally negatively charged proteins. Therefore the following separation happens solely by the size of the polypeptide chains in the polyacrylamide gel.
Recommended SDS-PAGE Buffer and Kits
Native PAGE:
Native, unfolded, and not-denatured proteins can be separated using this method. This method allows for the separation of proteins that are inaccessible by other methods. One example would be the separation of modified and unmodified proteins of the same kind (e.g. phosphorylated versus unphosphorylated state of a protein). Native PAGE can also be used to confirm biologically relevant conformations, like di-, tri-, or tetrameric forms of proteins (contrary to SDS-PAGE, which would separate the individual and denatured peptide chains). This method can also detect different complexes of different proteins.
The separation using native PAGE depends on a number of parameters such as the charge, size and 3D structure of the protein. A suitable buffer is needed to maintain the 3D folding of the protein. The applicability of the buffer depends on the isoelectric point and the charges of the protein.
Isoelectric Focusing:
This method builds on the fact that a protein has a specific charge at certain pH values. Depending on the pH the acidic and basic functional groups contribute by increasing or decreasing the total charge of the protein. The isoelectric point is defined as the the point where the total charge of the molecule is zero, because there is an equal amount of negative and positive charges in the molecule.
Special gradient gels are needed for isoelectric focusing as the pH changes from acidic to basic along a gradient within the gel. Due to an electric charge connected to the gel the protein travels to the point in the gel where the charge of the gel equals that of the protein, and the total charge equals zero, i.e. the isoelectric point. Hence, this method is used to separate proteins by their charges, as well as to determine the isoelectric point of a target protein. The separation occurs due to the charge of the protein or by the number of basic- and acidic groups the protein contains.
The above-mentioned methods for gel electrophoresis of proteins can also be combined to separate proteins. The choice of methods depends on the specific requirements of the experiment.
Blotting
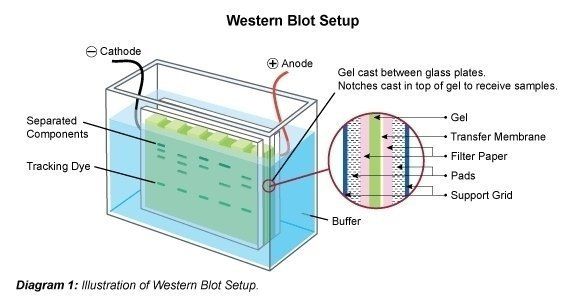
Following the separation of the protein mix the polypeptide bands are transferred to a membrane carrier. For this purpose the membrane is attached to the gel and this so-called sandwich is transferred to an electrophoresis chamber. It is possible that some of the SDS is washed out, and the protein partially re-naturates again, i.e. regains its 2D- and 3D structure. However, the applied electric charge causes the proteins to travel out of the gel vertically to the direction they traveled in on the gel, onto the membrane. The protein bands are thereby bound to the membrane. The "blotted" bands are now available to be treated further (e.g. for detection of specific proteins with specific antibodies).
Immunodetection
The identification of specific antibodies is possible after the separation and blotting of proteins. Specific antibodies (mono- or polyclonal) bind to "their" band of proteins. Unspecifically binding antibodies are removed by washing with detergent-containing buffers. Additionally, unspecific binding pockets can be blocked before the addition of specific antibodies.
Primary antibodies are usually applied first, which are then recognized by a secondary antibody. The secondary antibody is conjugated with colour, radioactivity or an enzyme for detection. Biotin-conjugated antibodies are also used for this purpose.
Recommended Blocking Buffers & Lysis Buffers for Western Blotting
It can occasionally be advantageous to use polyclonal primary antibodies as such antibodies recognize several epitopes, contrary to monoclonal antibodies that are restricted in their binding affinity. After immunodetection it is possible to strip the antibody off the membrane for further analysis with other antibodies (e.g. in order to detect other specific antibodies from the protein mixture under investigation).
Analysis of the western blot is then carried out using a variety of different imaging systems (e.g. luminescence, color reaction, autoradiography).
The identification of a protein of interest in a Western blot relies among other factors on its molecular weight. To this end, the electrophoretically separated proteins in the sample are compared with a molecular weight standard of a known composition.
Recommended Markers & Controls for Western Blotting
Why Western Blotting (Immunoblot)?
The western blotting (immunoblot) method entails various advantages as compared to other immunosorbent assays (ISAs), like for example ELISA.
Western blotting (immunoblot) expands on the idea of ELISA by allowing separation of the protein mix by size, charge, and/or conformation. The described method of stripping allows for the detection of several targets, contrary to ELISA where only one protein can be detected. As the gel electophoreis of proteins separates the proteins into bands, one can determine the size of the target protein/polypeptide. It is also possible to (semi-)quantify the protein of interest by running an internal quantity standard in parallel with the samples in the gel. Similarly, the protein content of the samples can be compared ("sample A contains more protein than sample B").
A disadvantage of western blotting (immunoblot) is that it is time-consuming (compared to ELISA) and has a high demand in terms of experience of the experimenter. Additionally, it requires optimizing the experimental conditions (i.e. protein isolation, buffers, type of separation, gel concentration, etc.).
There are many different types and methods for western blotting (immunoblot). Hence, it covers very different topics and applications.
Popular Antibodies for Western Blotting
Resources
Isotype Controls for Western Blotting
TrueBlot® Antibodies, Kits, Tools
Western Blotting in brief - The most important FAQs
What is Western Blotting?
Western blotting is a laboratory method for the detection and characterization of proteins in a sample. Western blotting, also known as immunoblotting, is a widely used laboratory technique in molecular biology and biochemistry. It is employed to detect specific proteins within a complex mixture of proteins extracted from cells or tissues. The technique involves a series of steps that begin with the separation of proteins based on their size using gel electrophoresis. The proteins are then transferred onto a membrane and subsequently probed with specific antibodies that bind to the target protein of interest. Through a series of chemical reactions, the bound antibodies are visualized, allowing for the identification and quantification of the protein. Western blotting provides valuable information about protein expression, post-translational modifications, and protein-protein interactions, making it an indispensable tool in biological research and clinical diagnostics.
What is Western Blotting used for?
Western blotting is used to identify proteins of interest in complex mixture of proteins. It is commonly used in molecular biology to determine protein expression and detect the target proteins in biological samples.
How does the Western Blotting method work?
In a Western blot, proteins are first separated by size using gel electrophoresis and then transferred to a nitrocellulose or PVDF membrane. The membrane is then incubated with a primary antibody that specifically binds to the target protein of interest. After washing off any unbound primary antibody, the membrane is incubated with a secondary antibody that recognizes and binds to the primary antibody. The secondary antibody is usually conjugated with a detection molecule, such as an enzyme or fluorescent tag, that generates a signal that can be visualized.
What is the difference between a reducing and a native Western Blot?
In a reducing or denaturing Western blot, proteins are analyzed after they have been denatured and reduced to break their disulfide bonds. This results in a protein sample that is no longer in its native state but allows for the detection of individual subunits or monomers. Usually, the electrophoresis buffer contains SDS to evenly coat the denatured proteins and keep them linearized so that they are separated based on its molecular weight. A native Western blot is used to analyze proteins in their native state without disrupting their structure or interactions. It is typically performed under non-reducing conditions, meaning that the disulfide bonds between cysteine residues are not reduced. Their structure and possibly interactions with other proteins remain intact and influence the migration in during electrophoresis.
Reducing Western blots are commonly used to detect a particular protein or subunit of a protein complex. Native Western blots are used to analyze intact proteins or protein-protein interactions.
What materials and reagents are needed for a Western Blot?
For a Western blot one needs a protein sample, polyacrylamide gel, a nitrocellulose or PVDF transfer membrane, transfer and blocking buffer, a primary and a secondary antibody, detection reagents such as ECL substrate, and equipment for gel electrophoresis and membrane transfer. Additional reagents such as reducing agents, loading buffers, and molecular weight markers may also be required depending on the specific experiment.
How do you prepare samples for Western Blot?
For a reducing Western blot, samples are dissolved in a Laemmli buffer and then boiled to assure that all the protein in the sample is denatured. They mixture is then rapidly cooled down on ice before loading to keep proteins in their denatured, linear form.
To prepare samples for a native Western blot, the sample is usually mixed with a native sample buffer that contains a non-ionic detergent, such as Triton X-100, to solubilize the proteins without disrupting their native structure or interactions. Coomassie blue can be used in native Western blotting to bind and coat proteins during electrophoresis (blue-native PAGE).
What type of gel should be used for Western Blot?
Polyacrylamide gel is the most commonly used gel for Western blotting. Denaturing SDS-PAGE is typically used for separating proteins based on their molecular weight. SDS-PAGE involves the use of a discontinuous buffer system with two gels - a stacking gel and a resolving gel. The stacking gel has a low percentage of acrylamide and serves to concentrate and focus the protein sample into a narrow band, while the resolving gel has a higher percentage of acrylamide and separates the proteins based on their size.For reducing Western blotting, a reducing agent such as beta-mercaptoethanol or dithiothreitol (DTT) is often added to the sample buffer to break the disulfide bonds between cysteine residues and fully denature the proteins.
For native Western blotting, the gel is prepared under non-denaturing conditions, and the disulfide bonds are not reduced. This is typically done using a "native" gel system or "blue-native" gel system (BN-PAGE), which uses a non-ionic detergent such as Triton X-100 to maintain the native conformation of the proteins.
How are proteins transferred to the membrane?
Proteins are most commonly transferred from the polyacrylamide gel to a blotting membrane through electroblotting. In a tank blot (wet transfer), the gel is sandwiched between two pads or sponges soaked in transfer buffer, and a membrane such as nitrocellulose or PVDF is placed on top of the gel. The stack is then placed into an electrophoresis chamber, and a current is applied to transfer the proteins from the gel onto the membrane. The transfer buffer typically contains a buffer system that maintains a pH and ionic strength that supports protein transfer, along with methanol or ethanol to enhance transfer efficiency. In a "semi-dry transfer" filter paper pads soaked in transfer buffer surround the gel and membrane. The sandwich is placed in direct contact with two electrodes through which an electric field is applied.
Subsequently to the transfer, the membrane is blocked to prevent non-specific binding of detection antibodies and then incubated with primary and secondary antibodies specific to the protein of interest. The bound antibodies are then detected by chemiluminescence or other detection methods, and the signal is captured using X-ray film or a digital imaging system.
How can nonspecific binding be reduced in the Western Blot? How does membrane blocking occur and why is it important?
After transfer of the proteins from the electrophoresis gel to the blotting membrane, the membrane is incubated with a blotting buffer containing protein such as BSA or milk powder. These proteins homogenously coat the membrane to block non-specific binding of the detection antibodies. Blocking the membrane prevents non-specific binding and reduces background signals.
Which antibodies are used for Western Blotting?
A primary antibody specific for the protein of interest or alternatively a protein tag is used for binding to the protein. A secondary antibody that binds to the primary antibody is typically used for detection. Frequently, the secondary antibody is conjugated to an enzyme like horseradish peroxidase (HRP) that transforms a substrate such as ECL for detection of the antibody-labeled protein band on the blotting membrane.
Primary antibodies recognize specific proteins, while secondary antibodies bind to the primary antibodies and produce a detectable signal. A labeled secondary antibody can be used with primary antibodies against different proteins of interest and it can be used to amplify the signal. It is also possible to directly label the primary antibody to avoid a second incubation and the associated wash steps.
How are protein bands detected on the membrane?
Protein bands are visualized through the use of detection reagents, such as chemiluminescent substrates that are transformed by an enzyme such as HRP. The produced chemiluminescent signal can then be captured using film or a digital imaging system.
How is the amount of protein quantified in the Western Blot?
Quantification of proteins via Western blot is semi-quantitative. Due to its upper and lower detection limits Western blot is primarily a qualitative method. The amount of protein can be estimated in Western blotting by measuring the intensity of the protein signal on the membrane using densitometry. This involves using a specialized instrument such as a scanner or imaging system to capture the signal intensity of the protein bands on the membrane, typically as a digital image. The image can then be analyzed using software that measures the signal intensity of each band, and this value can be compared to a standard curve or control sample to quantify the amount of protein. Alternatively, some Western blotting systems use a pre-stained molecular weight ladder or a reference protein in the sample as an internal control to estimate the protein amount based on its relative intensity compared to the control.
How can specific binding be detected in the Western Blot? How is the quality and reproducibility of Western blot data ensured?
Specific binding can be confirmed by comparing controls positive and negative controls, different primary antibody against the same protein of interest, and orthogonal methods such as chromatography, mass-spectrometry, or flow cytometry. Reproducible results across biological and technical replicates ensure the quality of the data.
What are the advantages of Western Blotting compared to other protein analysis methods?
Western blotting allows for high specific protein identification, semi-quantitative analysis, the ability to examine multiple proteins in one sample, and depending on the experimental layout the detection of protein interactions. Compared to other protein-focused methods such as chromatography, flow cytometry, or mass spectrometry, Western blot is inexpensive, easily accessible, and fast.
What are typical problems and challenges in Western Blotting and how can they be solved?
Typical problems in Western blotting include non-specific binding and background signals, weak signal and difficulty in detecting lowly expressed proteins, and variability between experiments. These can be addressed by optimizing antibodies, blocking methods, and protocol parameters, and by using internal controls.
Are there alternative methods to western blotting?
Yes, there are alternative protein analysis methods such as ELISA, mass spectrometry, protein arrays, flow cytometry, and immunohistochemical techniques that can be applied depending on the application.
What are the main applications of Western Blotting in research or clinical diagnostics?
Western blotting is a versatile technique that allows detection of a particular protein in a sample. Therefore, it can be applied to a wide range of research and clinical applications, making it an essential tool for many scientists and clinicians. Some of the main applications of Western blotting are disease diagnosis by detecting specific markers, antibody validation for downstream applications such as immunohistochemistry or flow cytometry, detecting antibodies in serum samples to assess vaccine efficacy, or quality control in biosimilars production to ensure consistency.

Goal-oriented, time line driven scientist, proficiently trained in different academic institutions in Germany, France and the USA. Experienced in the life sciences e-commerce environment with a focus on product development and customer relation management.
Go to author page



