CD Marker Panel
Martin HessCD is an abbreviation “for cluster of differentiation”. CD molecules are cell surface markers which are very useful for the identification and characterization of leukocytes and the different subpopulations of leukocytes. The HLDA (Human Leukocyte Differentiation Antigens) workshop, which started in 1982, developed the CD nomenclature and has maintained the list of CD Markers ever since. The initial idea behind the CD nomenclature was the classification of many different monoclonal antibodies against cell surface molecules of leukocytes which had been generated by different laboratories around the world.
The number of CD markers has grown constantly and was expanded to other cell types. Today there are more than 320 CD clusters described in humans. For more information and a comprehensive list of CD markers please visit www.hcdm.org.
CD Marker - mAbs against Surface Molecules of Leukocytes
CD Markers are especially useful for identification of leukocyte population using flow cytometry. Our resource page What is flow cytometry? provides a short introduction into flow cytometry. The great advantage of flow cytometry is that it allows for the simultaneous detection of several markers on a single cells at the very same time. With a modern flow cytometer 8-10 different colors can easily be measured in one sample, the most advanced cytometers can even measure up to 18 channels at once.
Most common CD Markers for Flow Cytometry
| Cell Type | Human | Mouse | Rat | Cow | Horse | Pig | Dog | Monkey/ Primate |
| Leukocytes | ||||||||
| T-Cells (General) | ||||||||
| T-Helper Cells | ||||||||
| Cytotoxic T-Cells | ||||||||
| Natural Killer Cells | ||||||||
| B Cells | ||||||||
| Dendritic Cells | ||||||||
| Monocytes / Macrophages | ||||||||
| Granulocytes | ||||||||
| Hematopoetic Stem Cells | ||||||||
| Platelets | ||||||||
| Erythrocytes | ||||||||
| Endothelial Cells |
*(links in bracket indicate that we cannot guarantee that the mentioned markers are the best defining marker of that cell population in the specific species)
Frequently used CD Marker Antibodies
- (8)
- (5)
- (8)
- (4)
- (8)
- (3)
- (6)
- (3)
- (4)
- (3)
- (2)
- (10)
- (1)
- (7)
- (2)
- (2)
- (8)
- (1)
- (9)
- (4)
Determination of Leukocyte Populations with CD-Markers
When using flow cytometry to determine certain leukocyte population usually a combination of several CD markers is used to determine a subpopulation, since one single marker usually falls short of defining the entire population. Usually research generate gating trees when evaluating flow cytometry experiments.
A very illustrative example is the distribution of CD8. While CD8 is the most prominent marker and namesake for CD8 positive (CD8+) cytotoxic T cells, it is also expressed by natural killer cells (NK-cells) and dentritic cells (DCs). If all CD8+ cells were gated from a leukocyte population of a blood sample the gated population would consist of CD8+ T cells, NK cells and DCs.
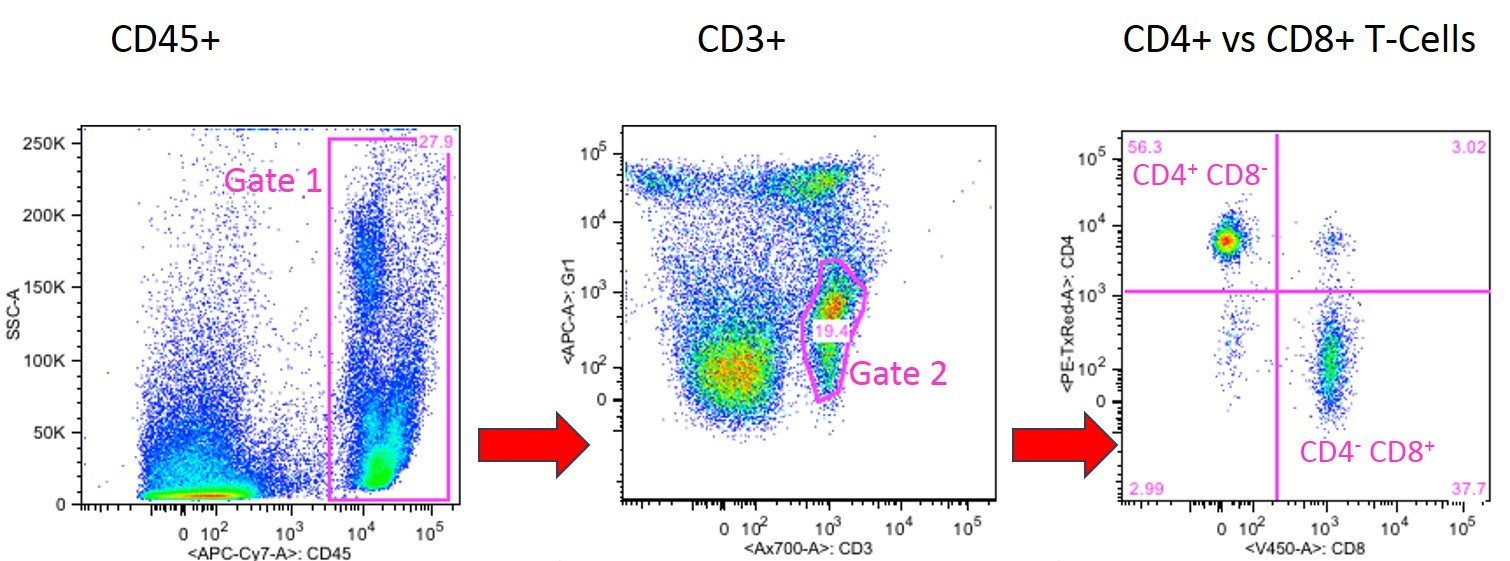
In the following experiment the ratio of CD4+ T helper vs. CD8+ cytotoxic T cells was determined. First all CD45+ cells (general leukocyte marker) of the peripheral blood sample were gated (Gate 1).
In the next panel all CD45+ cells of Gate 1 were blotted for their expression of CD3. CD3 is the most common T cell marker which is only expressed by T cells and not by other CD8+ cells such as NK cells or DC. By gating for CD3+ cells it is guaranteed that no CD3- but CD8+ cells (e.g. NK cells or DCs) are counted as CD8+ T cells.
The CD3+ T cell population can be further differentiated into cytotoxic CD8+ T cells and into CD4+ T helper cells as shown in panel2 where the CD3+ cells of Gate 2 were blotted for their expression CD4 and CD8. CD4+ CD8- T helper cells are found in the upper left quadrant and CD4- CD8+ cytotoxic T cells are shown in lower right quadrant of panel 3.
If needed a researcher interested in different T helper Population such as TH1, TH2, TH9, TH17 and Treg would stain for the respective extra or intracellular markers of those subpopulation.
CD-Markers - Some Important Questions and Answers
What are CD clusters and what are they used for?
CD clusters (clusters of differentiation) are surface marker molecules found on cells. They are used in immunology to identify and characterize different cell types.
Which CD clusters are present on a particular cell population?
Each cell type has a specific set of CD markers and can be determined by them. Cell types are defined based on their specific endowment with CD markers. For example, all leukocytes carry CD45. Cells that are positive for CD3 and CD8 are called cytotoxic T cells. Different developmental stages of a cell type can also be detected using the specific set of CD markers. Identifying CD clusters on a cell population requires the use of techniques such as flow cytometry.
How does CD cluster expression change during development or in specific disease states?
The expression of CD clusters can change during cell development: If cells change due to disease, so does the specific set of CD markers that scientists can use to detect the cell changes. Certain disease states can thus have an impact on the expression of CD clusters. To study such changes, scientists can perform comparative analyses between different developmental stages or disease states, for example, by comparing tissue samples or cell cultures.
What are the functions of specific CD clusters on cells?
The functions of CD clusters on cells can be diverse. Some CD clusters serve as receptors for ligands and can activate signaling pathways that affect cell behavior and function. Other CD clusters are involved in cell adhesion or immune recognition.
How can we quantify the expression of CD clusters?
The expression of CD clusters can be quantified by various experimental methods. One commonly used method is flow cytometry, which uses specific antibodies to detect the expression of CD clusters on single cells. Alternatively, techniques such as single cell RNA sequencing can be used to analyze the expression of CD clusters on a genome-wide level.