BAD Antibodies
Your search for reliable BAD antibodies ends here. BAD, known by aliases such as badb, BAD, Bad, is an integral part of our antibody range. Whether you're working with Human, Mouse, Rat, Rabbit, Monkey, or other species, our range of BAD antibodies offer precise detection across diverse samples. These specialized antibodies are tailored for various scientific applications like ELISA, WB, DB, IHC, IF (p), providing you with options like polyclonal, recombinant, and monoclonal antibodies, sourced from different host species such as Rabbit, Mouse, Rat. The efficacy of our antibodies is well-established, demonstrated through multiple methods.
Detailed information, including references, images, and validations by other customers, can be found on each product page. Should you require assistance in finding a specific product, our customer service team is ready to assist. Utilize our BAD antibodies in your research endeavors for dependable BAD detection.
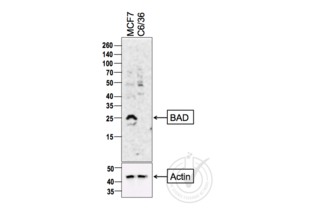
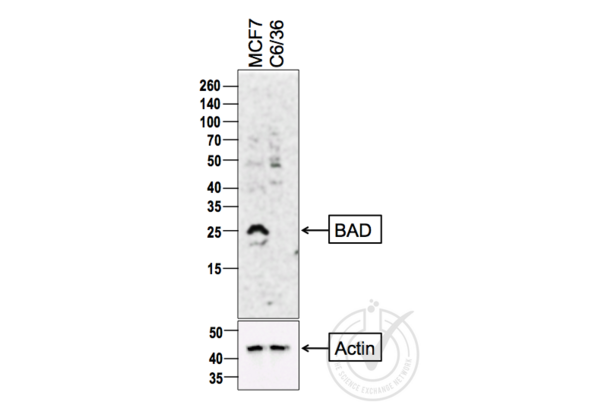 BAD antibody (AA 101-204) (ABIN674709)
BAD antibody (AA 101-204) (ABIN674709)
BAD Reactivity: Human, Mouse, Rat ELISA, WB, IHC (p), IF (p), IF (cc), FACS, ICC, IHC (fro) Host: Rabbit Polyclonal unconjugated
BAD Reactivity: Human WB, IHC, ICC, IP Host: Rabbit Polyclonal unconjugated
BAD Reactivity: Human ELISA, WB, FACS Host: Mouse Monoclonal 1G5B3 unconjugated
BAD Antibodies by Host
Find BAD Antibodies with a specific Host. The Host listed below are among those available. Click on a link to go to the corresponding products.
BAD Antibodies by Clonality
Find available monoclonal or polyclonal BAD Antibodies. Click on a link to go to the corresponding products.
Popular BAD Antibodies
- (3)
- (1)
- (8)
- (5)
- (4)
- (4)
- (3)
- (3)
- (2)
- (3)
- (5)
- (5)
- (3)
- (3)
- (3)
- (2)
- (2)
- (2)
Latest Publications for our BAD Antibodies
: "Biological effects of whole Z.Officinale extract on chronic myeloid leukemia cell line K562." in: Gene, Vol. 692, pp. 217-222, (2019) (PubMed).: "GABA A receptor π subunit promotes apoptosis of HTR-8/SVneo trophoblastic cells: Implications in preeclampsia." in: International journal of molecular medicine, Vol. 38, Issue 1, pp. 105-12, (2017) (PubMed).
: "Photodynamic Therapy Using Indolines-Fused-Triazoles Induces Mitochondrial Apoptosis in Human Non-Melanoma BCC Cells." in: Anticancer research, Vol. 37, Issue 10, pp. 5499-5505, (2017) (PubMed).
: "Expression and localization of angiopoietin family in buffalo ovarian follicles during different stages of development and modulatory role of angiopoietins on steroidogenesis and survival of cultured ..." in: Theriogenology, Vol. 86, Issue 7, pp. 1818-33, (2016) (PubMed).
: "Expression and localization of angiopoietin family in corpus luteum during different stages of oestrous cycle and modulatory role of angiopoietins on steroidogenesis, angiogenesis and survivability ..." in: Reproduction in domestic animals = Zuchthygiene, Vol. 51, Issue 6, pp. 855-869, (2016) (PubMed).
: "Involvement of caspases and their upstream regulators in myocardial apoptosis in a rat model of selenium deficiency-induced dilated cardiomyopathy." in: Journal of trace elements in medicine and biology : organ of the Society for Minerals and Trace Elements (GMS), Vol. 31, pp. 85-91, (2015) (PubMed).
: "Analysis of protein-protein interactions in cross-talk pathways reveals CRKL protein as a novel prognostic marker in hepatocellular carcinoma." in: Molecular & cellular proteomics : MCP, Vol. 12, Issue 5, pp. 1335-49, (2013) (PubMed).
: "Loss of Bad expression confers poor prognosis in non-small cell lung cancer." in: Medical oncology (Northwood, London, England), Vol. 29, Issue 3, pp. 1648-55, (2012) (PubMed).
: "ZIC1 is downregulated through promoter hypermethylation, and functions as a tumor suppressor gene in colorectal cancer." in: PLoS ONE, Vol. 6, Issue 2, pp. e16916, (2011) (PubMed).
: "BAD contributes to RAF-mediated proliferation and cooperates with B-RAF-V600E in cancer signaling." in: The Journal of biological chemistry, Vol. 286, Issue 20, pp. 17934-44, (2011) (PubMed).
Aliases for BAD Antibodies
BCL2-associated agonist of cell death b (badb) AntibodiesBCL2 associated agonist of cell death (BAD) Antibodies
BCL2-associated agonist of cell death (Bad) Antibodies
AI325008 Antibodies
bad Antibodies
BAD Antibodies
BBC2 Antibodies
Bbc2 Antibodies
BCL2L8 Antibodies
fa01b12 Antibodies
wu:fa01b12 Antibodies
wu:fa96d04 Antibodies
Did you look for something else?
- Baculoviral IAP Repeat-Containing 7 Antibodies
- Baculoviral IAP Repeat-Containing 5 Antibodies
- BACH2 Antibodies
- BACH1 Antibodies
- BACE2 Antibodies
- BACE1 Antibodies
- BAAT1 Antibodies
- BAAT Antibodies
- BAALC Antibodies
- B9D2 Antibodies
- B9D1 Antibodies
- B7-H6 Antibodies
- B4GALT7 Antibodies
- B4GALT6 Antibodies
- B4GALT5 Antibodies
- B4GALT4 Antibodies
- B4GALT3 Antibodies
- B4GALT2 Antibodies
- B4GALT1 Antibodies
- B4GALNT2 Antibodies
- BAFF Antibodies
- BAG1 Antibodies
- BAG2 Antibodies
- BAG3 Antibodies
- BAG4 Antibodies
- BAG5 Antibodies
- BAGE3 Antibodies
- BAGE4 Antibodies
- BAGE5 Antibodies
- BAHD1 Antibodies
- BAI1 Antibodies
- BAI1-Associated Protein 2-Like-1 Antibodies
- BAI2 Antibodies
- BAI3 Antibodies
- BAIAP2 Antibodies
- BAIAP2L2 Antibodies
- BAIAP3 Antibodies
- BAK1 Antibodies
- Band 3/AE1 Antibodies
- BANF1 Antibodies




